Search This Blog
Saturday, October 31, 2020
NOVO NORDISK Gets a Buy rating from Bernstein
AbCellera Enters Pact with Kodiak Sciences to Develop Eye Disease Antibodies
AbCellera and Kodiak Sciences have announced a new partnership to create and develop therapeutic antibodies for ophthalmologic indications. The two companies originally entered a partnership in August 2016.
Under the new deal, AbCellera will leverage its full stack, artificial intelligence-powered antibody discovery platform to discover and identify a number of antibodies against targets selected by Kodiak. Kodiak focuses on products to treat and prevent blindness. Kodiak’s ABC Platform utilizes molecular engineering to combine antibody-based and chemistry-based therapies.
“Kodiak was one of AbCellera’s very first partners, and we are proud to expand our collaboration to support their innovative team in the fight against retinal diseases that cause blindness,” said Carl Hansen, AbCellera’s president and chief executive officer. “We are committed to making state-of-the-art antibody discovery broadly available to partners, empowering them to bring new therapies to patients quickly.”
Although financial terms were not disclosed, Kodiak is paying AbCellera research and development payments, various milestones and royalties on net sales.
Earlier this month, Kodiak reported that the first patients had been treated in three pivotal Phase III trials, GLEAM, GLIMMER and BEACON of KSI-301, its anti-VEGF antibody biopolymer conjugate. GLEAM and GLIMMER are in treatment naive patients with diabetic macular edema, and BEACON is in macular edema due to retinal vein occlusion.
In addition, they indicated that recruitment of patients in the U.S. for its DAZZLE Phase IIb/III trial of KSI-301 in treatment-naïve wet (neovascular) age-related macular degeneration is now complete, while recruitment in Europe is ongoing.
KSI-301 is an anti-VEGF therapy designed to maintain potent and effective drug levels in eye tissues for longer than current drugs.
In late September, AbCellera entered into a multi-year, multi-target research collaboration and license deal with IGM Biosciences. For that pact, AbCellera will create panels of antibodies for multiple therapeutic targets selected by IGM. IGM will have the rights to develop and commercialize the antibodies that come out of it.
At the time, Fred Schwarzer, chief executive officer of IGM Biosciences, said, “We believe that AbCellera’s technology will assist us in rapidly identifying the best antibodies against a large number of agonist targets, as well as other historically difficult targets, for both oncology and immunology applications. Engineered IgM and IgA antibodies may be able to overcome some of the limitations of the current IgG-based therapeutics, and this partnership demonstrates the depth of our commitment to ensuring that the potential of our IgM and IgA technology platform is fully realized in areas of high unmet medical need.”
Recently, AbCellera has been in the news for LY-CoV555, the neutralizing antibody Eli Lilly and Company is developing to prevent and treat COVID-19. The antibody came out of a collaboration between the two companies. LY-CoV555 is a potent, neutralizing IgG monoclonal antibody directed against the spike “S” protein on SARS-CoV-2, the virus that causes COVID-19.
In mid-September, Eli Lilly and Company announced interim proof-of-concept data from its BLAZE-1 Phase II clinical trial of LY-COV555 (bamlanivimab), its neutralizing antibody therapy for COVID-19. That study enrolled mild-to-moderate COVID-19 patients who had been diagnosed in the outpatient setting. This study is continuing.
However, a separate study of the antibody in hospitalized patients has been halted. About two weeks ago an independent Data Monitoring Committee paused enrollment in this trial because of a potential safety issue. The National Institute of Allergy and Infectious Diseases (NIAID), the study sponsor, could not verify the safety issue, but their analysis also indicated there was little likelihood the antibody would help the hospitalized patients.
https://www.biospace.com/article/abcellera-and-kodiak-ink-ophthalmology-deal/
As coronavirus surges again, US mortality rates highest in rural counties
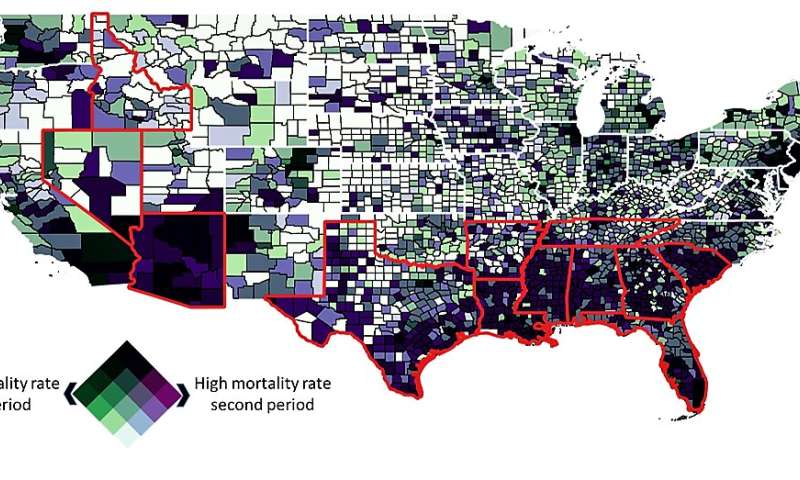
In parts of the country where COVID-19 is surging, residents of rural counties are dying from coronavirus at higher rates than those in urban areas, according to a new health policy brief by the University of Cincinnati.
Coronavirus infections are hitting rural parts of the United States previously spared, requiring a new approach to battling the pandemic, a multidisciplinary team of UC researchers said.
UC's Geospatial Health Advising Group compared mortality rates from COVID-19 in the early part of the pandemic when some states were locked down to more recent months when most schools and businesses reopened. They also compared urban and rural counties in states with high and low rates of infection.
Composed of health, geography and statistical modeling experts from the UC College of Pharmacy and the UC College of Arts and Sciences, the UC group formed in the spring to track the virus and provide guidance for Ohio's Department of Health.
Nationwide, urban areas have a higher rate of mortality from COVID-19 than rural areas. But in high-infection states where the virus is surging, the story is much different.
Rural concern
UC researchers warn that rural areas in these states have higher mortality rates from COVID-19 compared to cities. The epidemic shifted from the Northeast and mid-Atlantic regions to southern states that saw a surge in cases.
This is cause for concern for several reasons. Rural areas in the United States have fewer health care resources than cities. More than 16 million Americans live in counties with no surgical or intensive-care hospitals. And another 4.7 million Americans live in counties with no general medical or surgical beds at all.
The population in rural areas also skews much older than that of urban areas, according to the U.S. Census Bureau, which is a troubling consideration for a respiratory illness that is most dangerous to older patients.
Both factors make rural areas especially vulnerable to COVID-19.
"In mid-July hospitalizations were declining. Now they're going back up," said Diego Cuadros, assistant professor of geography. "It's beginning to saturate the health care system, which is what we want to avoid."
National recommendations
UC's Geospatial Health Advising Group offered several recommendations:
- Urging government health agencies to recognize the disproportionate impact COVID-19 is having on rural areas when drafting county-level policies to address the pandemic.
- Making residents in rural areas aware of the risks they face so they can take personal measures to protect themselves.
- Improving health care resources in rural areas through health partnerships, particularly for critical access hospitals.
Some countries like Japan and South Korea have done a better job than others of employing contact tracing and preventive measures such as masks and social distancing to starve the virus of hosts. But it's harder to isolate a virus in bigger countries with many points of entry.
There has been strong resistance in the United States and some European countries against lockdowns, social distancing and facemasks.
"We're having the same national conversation now that we had in the spring," Cuadros said. "People really want to go back to normal and it's not happening anytime soon. It's hard to convince people that they need to take precautions."
UC College of Medicine student Dillon Froass condensed UC's data, maps and pages of analysis to draft the brief as part of his fourth-year capstone project.
"I wanted to do something relating to the COVID-19 pandemic," he said. "It's been a great opportunity. You feel like you're helping people. Policymakers can learn more from this so it will make a difference."
Froass, a University Honors student and Darwin T. Turner scholar, plans to apply for medical school after taking a gap year to work at a hospital.
Froass said his college experience hasn't been hampered much by recommended social distancing. UC has had comparatively few infections, suggesting students are taking the risk of infection seriously. To date: UC has seen 742 cases out of a student body of 46,388. UC set a new record for enrollment this fall.
"I've been lucky that most of my classes have had in-person opportunities," he said. "Missing football games is a bummer, but it's well worth it if it means saving lives."
https://medicalxpress.com/news/2020-10-coronavirus-surges-mortality-highest-rural.html
COVID-19 patients infect half of household: CDC
People who develop COVID-19 infect around half of their household members, with adults only slightly more likely than children to spread the virus, a US government study said Friday.
The paper by the Centers of Disease Control and Prevention (CDC) is the latest to attempt to quantify the household transmission rate of the disease, with previous research varying widely but generally suggesting that adults are bigger drivers than children.
The new research by the CDC involved finding cases of "index" or initial patients with lab-confirmed coronavirus infection in Nashville, Tennessee, and Marshfield, Wisconsin, starting in April 2020.
Both the index patients and their household members were trained remotely to complete symptom diaries and obtain self-collected specimens, which were either nasal swabs only or nasal swabs and saliva samples, for 14 days.
A total of 191 enrolled household contacts of 101 index patients reported having no symptoms on the day of their index patient's illness onset.
In the follow-up period, 102 of the 191 contacts had SARS-CoV-2 positive tests, for a "secondary infection rate" of 53 percent.
The secondary infection rate when index patients were over 18 was 57 percent, which fell to 43 percent when the index patient was under 18.
Overall there were far fewer children index patients than there were adults: 20 compared to 82, which makes it harder to generalize the results for under-18s.
In terms of household characteristics, the median number of members per bedroom was one, 69 percent of index patients reported spending four or more hours in the same room with one or more household member the day before, and 40 percent the day after illness onset.
Forty percent of index patients reported sleeping in the same room with one or more household members before illness onset and 30 percent after illness onset.
Higher than reported
Interpreting the findings, the authors of the paper wrote: "In this ongoing prospective study that includes systematic and daily follow-up, transmission of SARS-CoV-2 among household members was common, and secondary infection rates were higher than have been previously reported."
"Substantial transmission occurred whether the index patient was an adult or a child," they added.
Another important finding of the study was that fewer than half of household members with confirmed infections reported symptoms at the time infection was first detected, and many reported no symptoms throughout seven days of follow-up.
This underscores the potential for transmission for asymptomatic secondary contacts.
Other studies carried out abroad have at times found lower household infection rates.
The CDC said this might be because those studies didn't have enough follow-up, or because those patients isolated in facilities outside their houses or applied more stringent mask use.
It recommended that people who think they might have COVID-19 should isolate themselves from others in their household, including sleeping separately and using a separate bathroom if possible, and wear a mask.
People exposed should not delay isolating until their infection is confirmed by a test.
An important limitation of the study was that determining who the index patient was can be challenging.
When the calculations were changed to exclude 54 household members who had positive tests in specimens taken at enrollment, but whose results took some time to be confirmed, the overall secondary infection rate fell to 35 percent.
However, it's still thought more likely that the person who first developed symptoms is the index patient.
https://medicalxpress.com/news/2020-10-faster-wider-covid-households.html
Indonesia set to become hub for Chinese vaccines in Southeast Asia

Getting a vaccine has become the ultimate escape route from the pandemic with more than a million lives lost and billions more at risk from coronavirus.
Countries are willing to spend billions of dollars to obtain the COVID-19 vaccine, which is highly promising for the vaccine industry in coming years.
Indonesia's opportunity to become a center of the COVID-19 vaccine industry is wide open with an offer from China to make Southeast Asia's largest economy the hub for the production and distribution of Chinese vaccines in the region.
Chinese Foreign Minister Wang Yi said his country was willing to work with Indonesia to "promote the research and development, production and use of the vaccine […] in the region and even the world".
So far Indonesia has become a testing ground for a vaccine being developed by China's Sinovac Biotech Ltd. Sinovac will also co-operate with Indonesian pharmaceutical holding company Bio Farma to produce the COVID-19 vaccines.
This opportunity will not only give Indonesia the benefit of securing first access to the vaccines but also enable the country to cash in on the profits from vaccine production and distribution.
Gaining economic benefits
Having first access to COVID-19 vaccines could help Indonesia's stalled economy, which has stalled during the pandemic. The country is set to enter a recession marked by two consecutive quarters of negative economic growth.
Indonesian Finance Minister Sri Mulyani Indrawati has forecast the economy might dip to minus 2.9% in the third quarter of this year after contracting by 5.32% in the second quarter.
If people are vaccinated against the virus, economic activity can be restored and the economy would recover. The potential global income boost from coronavirus vaccines is up to US$9 trillion by 2025. For Indonesia, it is estimated a half-year delay in the delivery of COVID-19 vaccines will result in a massive economic loss of US$44 billion.
Not only does it save the Indonesian people as well the economy, if Indonesia can maximize its role as the hub of COVID-19 vaccines for Southeast Asia, the country may gain a share of the profits.
With a total population of 670 million, Southeast Asia is undoubtedly an attractive market. The value of the region's total vaccine imports was US$223 million in 2010 and almost quadrupled to US$859 million in 2019.
World Bank trade statistics show Indonesia is the biggest vaccine exporter in Southeast Asia. The country earned US$76.3 million from vaccine exports in 2010, which grew by 25.2% to US$95.5 million in 2019. Singapore and Thailand followed Indonesia with a combined export value of US$80.7 million in 2019.
The vaccine industry may also create new employment opportunities. Indonesia's annual manufacturing survey shows the vaccine industry created more than 1,500 jobs in 2018. The expansion of the industry will likely bring in more new jobs.
That figure is relatively small compared to the 3.5 million jobs lost during the pandemic. Yet the vaccine industry may bring other economic benefits.
Under the agreement with China, Indonesia becomes not only the main distributor of Chinese vaccines for the region but also the producer. This means Indonesia will save money on vaccine imports.
If the country wants to achieve herd immunity and stop the pandemic, it must vaccinate at least 50% of its population. Sinovac's vaccine price is tagged at US$60 for its full double dose. Therefore, the country needs to spend at least US$8.2 billion to vaccinate 50% of the population of 273.5 million. This amount is equal to more than twice our trade deficit last year of US$3.2 billion.
The number is just an estimate. The real total cost might be higher, depending on the vaccine's efficacy and the type of the vaccine (single or two-dose vaccine). If the vaccine is less effective, the number of people who need to be vaccinated will be higher than 50% to be able to achieve herd immunity. If a dual-dose vaccine is needed, the number of vaccines that has to be produced would double.
Reducing imports by producing COVID-19 vaccines locally will also help our trade balance. Indonesia is set to produce 250 million doses of vaccines by December. The production involves Sinovac as the vaccine provider, while Indonesia's Bio Farma will be responsible for vaccine packaging.
If Indonesia could sell its locally value-added COVID-19 vaccines to other Southeast Asian countries, it would generate income for the country.
The challenges
Although it promises many benefits, there is an enormous challenge to be overcome before Indonesia can benefit from becoming Southeast Asia's vaccine hub.
First is the mismatch between domestic production capacity and regional needs.
Bio Farma has a production capacity of 100 million vaccine doses a year. The firm is investing US$88.6 million to increase production capacity to 250 million doses a year in 2021. Considering each person will need at least two doses, its annual vaccine production would be only enough for 125 million people.
Even with the upgraded capacity, it would still take years to supply Southeast Asia's or even domestic needs. However, as the need for vaccines is urgent, production capacity may soon be increased further. It is predicted the capacity to produce COVID-19 vaccines won't be sufficient until 2024.
The next challenge is to increase the proportion of domestic content in COVID-19 vaccine production. Indonesia is only contributing to the packaging process, which is of relatively small value. In the future, Indonesia is expected to develop its own vaccines and the country should benefit from co-operation with China to enable knowledge and technology transfers from China to Indonesia.
With a supportive business environment and backing from China, the pandemic could be a blessing in disguise for Indonesia's vaccine industry to tap into the regional market.
https://medicalxpress.com/news/2020-10-indonesia-hub-chinese-vaccines-southeast.html