by Carly Mayberry via The Epoch Times (emphasis ours),
It’s been a year since four Department of Defense (DOD) whistleblowers found a sudden increase in various diseases in the Defense Medical Epidemiology Database (DMED), which coincided directly with the introduction of COVID-19 vaccinations. Now, new data shows more evidence.
That’s according to Lt. Col.Theresa Long, M.D., MPH, a board-certified aerospace medicine doctor and Army Brigade flight surgeon with specialty training as an aviation mishap investigator and safety officer, who was one of the four whistleblowers. Long’s background has uniquely equipped her to recognize what she described as “unusual diagnoses and alarming trends only after the introduction of the COVID-19 vaccinations.”
Sharp Increase in Serious Harm Reports in Pilots: DOD Data
Long said what she has now found has led her to file yet another whistleblower complaint with Sen. Ron Johnson’s (R-Wis.) office. She described this data as “more alarming DMED data” after she “went back into the ‘fixed’ DMED again to look for signals of harm for Army aviation.”
“What I found was a clear signal, that something in 2021 changed the health of service members,” Long told The Epoch Times. She said these signals were consistent with those in the Vaccine Adverse Event Reporting System (VAERS) reports. But unlike VAERS reports, DMED data showed spikes in the number of diagnoses “made by a healthcare professional within the DOD on service members.”
According to the Military Health System, the DMED provides remote access to a subset of data contained in the Defense Medical Surveillance System (DMSS). The DMSS contains up-to-date and historical data on diseases and medical events (including reportable events) and “is available to authorized users such as U.S. military medical providers, epidemiologists, medical researchers, safety officers or medical operations/ clinical support staff for surveying health conditions in the U.S. military.”
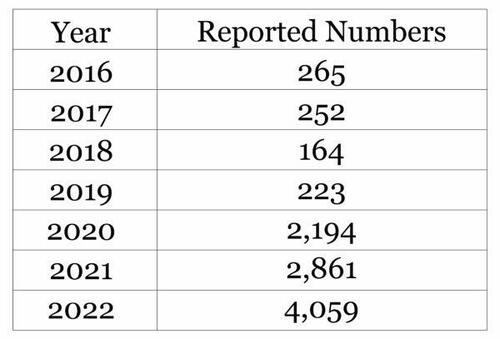
“After querying all pilots across the DOD, for all-cause morbidity and mortality, I found a stunning increase in the number of reportable events, spiking from an average of 226 reportable events a year (2016-2019) to 4,059 reports in 2022,” she explained.
A DOD reportable event is any patient safety event resulting in death, permanent harm, or severe temporary harm—and all require a comprehensive systematic analysis and a follow-on corrective action implementation plan report.
“The point is there is a statistically significant increase in death, permanent harm, or severe temporary harm in young healthy fit pilots,” she continued.
Such injuries were more obviously shown in this population. Because aviation pilots are required to have a superior level of health and fitness, and their health conditions are under more strict monitoring, according to Long.
What spurred Long on to pull this second round of data was when she learned the Federal Aviation Administration (FAA) had quietly made changes to the acceptable parameters of PR intervals (representative of the first part of a heartbeat, measured in seconds or milliseconds) on electrocardiograms of pilots. The FAA didn’t respond with research and data to support their decision, according to Long.
Those actions led to the press release dated Jan. 27, 2023 from Johnson in a letter to the FAA, where he stated the following details:
“Based on data from the Defense Medical Epidemiology Database, the whistleblower [Theresa Long] reported that the total number of disease and injuries [reportable events] in pilots across the DOD was 265 in 2016, 252 in 2017, 164 in 2018, 223 in 2019, 2,194 in 2020, 2,861 in 2021, and 4,059 in 2022.”
Johnson also told The Epoch Times these statistics “raise questions as to whether FAA has seen similar increases in disease and injuries in individuals in the aviation industry.”
Long noted that in the “post-glitch” DMED, the number of reportable events across the DOD had gone from a four-year average (2016-2019) of 40,813 to 110,000 in 2020 to over 200,000 in 2022.
“Some would ask why the numbers start increasing in 2020, you have to remember the Pfizer/DOD study with 43,448 participants started on July 27, 2020.”
Long emphasized that her opinions do not reflect those of the Army or the DOD.
Looking back, she said it was after being stonewalled for answers regarding adverse events from the COVID vaccine that she began performing queries in the DMED. She wanted to know if what she was seeing within her brigade were isolated anomalies or part of a wider disaster unfolding.
Whistleblowers First Report Discrepancies in DOD Data
It was in January of 2022 when Long, along with two other U.S. military doctors, Dr. Samuel Sigoloff and Special Forces flight surgeon Lt. Col. Peter Chambers, and Army Public Health Officer 1 Lt. Mark Bashaw first blew the whistle on the DOD. Together, they filed the initial whistleblower complaints regarding the DMED data, which showed an inordinate amount of negative health-related conditions related to the vaccine.
The initial DMED data given to Johnson showed a massive rise in cases of anxiety, esophageal cancer, breast cancer, female infertility, miscarriages, HIV, acute myocarditis, and Bell’s palsy among other conditions after the vaccine was mandated for U.S. military members.
Long added that after the DMED data was presented, Moderna, the pharmaceutical and biotechnology company behind one of the COVID-19 vaccinations and its mRNA immune response technology, lost $140 billion of dollars in stock.
Yet, despite the alarming data coming directly from the DOD’s own $42 million medical surveillance database, the department’s official claimed that the discovery of the data was a “data glitch” and proceeded to take the database offline, supposedly “fixing” it.
As reported in The Epoch Times, the DOD claimed that the data in DMED was incorrect for the years 2016-2020, but the 2021 number was not affected. The corrected data saw the data for prior years increased, which made the 2021 data look normal.
After Long handed over the documents to DOD, it took officials 47 days to formulate a response to the data, only to explain it was a surprise to them.
Based on the previous DOD data, “the cluster of medical conditions represents a dramatic shift in the acuity of medical conditions we normally see,” said Long, noting that the data is “so catastrophic,” at the very least when those numbers came out, the military would reflexively pause everything and investigate.
“They didn’t pause anything and it took them [the DOD] a month to complete their sham investigation.” She said. “It’s a gross indictment and dereliction of duty.”
“We introduce a brand new drug into our very healthy population and the surveillance people aren’t even paying attention to their own $42 million-a-year system?” asked Long, who noted that during her 30 years in the Army, many of which she served as a doctor, she and other colleagues never heard of such a database provided by the system’s contractor Ussiant until 2019. “Don’t you think introducing a drug that was rushed to an entire fighting force would make it a top priority that the surveillance system is working?”
Long also asked why, if the DMED just had a “glitch” during the COVID pandemic, no one is being held accountable for this egregious medical surveillance system failure. Long’s attorney, Todd Callender, noted the DOD failed to produce a single expert IT witness that would testify under oath that the shocking data was just a “glitch.”
“So if the data was that alarming, why didn’t anyone in the Defense Health Agency (DHA) sound the alarm or catch the ‘glitch,’” she continued. “How did they not see this huge spike in serious medical problems?”
Another question arises as to why military doctors like Long have not received any communication regarding this spike in reportable events, which wasn’t just limited to pilots but also general officers and those in the Special Forces.
“I was notified to comb over our inventory after a risk management alert notification alerted me to two defective earplugs found at Fort Sill, Okla.” Long said, “But I can’t even get them to send out an alert saying ‘Hey your pilots might get myocarditis from the vaccine.’”
For this story, The Epoch Times reached out for comment from Director of Defense Lloyd J. Austin, the Office of the Surgeon General, and the U.S. Department of Health and Human Services for comment.
‘I Can’t Un-see the Things I’ve Seen’
These new developments come as more physicians and patients have spoken out about a growing number of vaccine injuries while the science and research literature has simultaneously validated their claims and concerns.
Long said she was not only ignored but received threats against her career after speaking up. That’s because no action was taken on the part of military leaders to fully investigate the number and scope of adverse medical events that she, Sigoloff, Chambers, and Bashaw initially brought to their attention.
“When I found the DOD data, they pulled my credentials and took all my patients off my schedule,” said Long, noting that only left her more time to thoroughly look into the data.
While Long continues to add to her count of personally witnessed vaccine injuries, she also waits for a response from government officials with her latest filing.
Since she first came forward, she has also given testimony to the Idaho Legislature and at the Alaska Medical Freedom Symposium. Appearing recently on Fox News’ Tucker Carlson Tonight, she spoke about the FAA’s change in health requirements that significantly broaden the electrocardiogram range for pilots and allows those with cardiac injury damage to fly.
“In the light of emerging and overwhelming data showing cardiac damage from COVID and COVID vaccines on cardiac muscle, I can’t imagine why they would make this move and I think it’s a question that really should be taken to Dr. Susan Northrup, senior flight surgeon for the FAA,” Long told Carlson.