Novartis’
Cosentyx hit the mark in treating patients with non-radiographic axial
spondyloarthritis (nr-axSpA). In a Phase III trial, Cosentyx showed a
significant and clinically meaningful reduction in disease activity for
patients versus placebo.
Data from the Phase III PREVENT trial
showed that Cosentyx (secukinumab) demonstrated a sustained response
and maintained a consistent safety profile at 52 weeks. At the end of a
year, treatment with Cosentyx met its primary endpoint of ASAS40, a
typical measurement for the disease. As Novartis noted, ASAS40 is
achieved when there is a measure of an improvement of at least 40% and
an improvement of at least 10 units on a 0–100 scale in at least three
of the following domains: Patient global assessment, Pain assessment,
Function according to the Bath Ankylosing Spondylitis Functional Index,
and Inflammation.
There are approximately 1.7 million patients with nr-axSpA in the United States and European Union, Novartis said.
No new safety signals were detected, the Swiss pharma giant announced Wednesday. Positive 16-week PREVENT data
were announced in the middle of September and submitted to European
Medicines Agency for approval in nr-axSpA. The company intends to file a
supplemental New Drug Application with the U.S. Food and Drug
Administration for Cosentyx in nr-axSpA. The data from the PREVENT trial
add to the five-years of clinical data
posted last year supporting the long-term efficacy and safety of
Cosentyx across ankylosing spondylitis, psoriatic arthritis and
psoriasis, the company said.
Full data from the PREVENT trial will be presented at a future
medical conference, Novartis said. Cosentyx is an interleukin-17A
(IL-17A) inhibitor. IL-17A is produced by various cells from both the
innate immune system (which can be triggered by mechanical stress) and
the adaptive immune system. Cosentyx has been approved to treat plaque
psoriasis, active psoriatic arthritis and ankylosing spondylitis, a form
of arthritis that affects the spine.
John Tsai, Novartis’ chief medical officer and head of global drug
development, said the data from the PREVENT trial are encouraging for
people living with nr-axSpA, a chronic form of arthritis that typically
affects the low back and buttocks, which has limited treatment options.
“It’s a great example of how we’re working to reimagine medicine to
help patients realize early relief from this disease,” Tsai said in a
brief statement.
Atul Deodhar, medical director of Rheumatology Clinics at Oregon
Health & Science University, and an investigator in the secukinumab
clinical trial program, said that if left untreated, axial
spondyloarthritis can have a significant negative impact on quality of
life. The positive results from the PREVENT trial show that Cosentyx
could be a potential new treatment option for these patients.
https://www.biospace.com/article/novartis-eyes-new-cosentyx-approval-after-positive-phase-iii-axial-spondyloarthritis-trial/
Search This Blog
Friday, October 4, 2019
Sarepta up 5% premarket on positive data from SRP-9003 trial
Sarepta Therapeutics (NASDAQ:SRPT) is up 5% premarket on announcing
nine-month functional results from three Limb-girdle muscular dystrophy
Type 2E (LGMD2E) clinical trial participants who received SRP-9003, an
investigational gene therapy intended to transduce skeletal and cardiac
muscle with a gene that codes for the full-length, native
beta-sarcoglycan protein.
In Cohort 1 of the study, three participants ages 4-13 were treated with an infusion of SRP-9003 at a dose of 5×1013vg/kg. Improvements in functional outcomes were observed at day 270 (nine months) for all three participants.
At Day 270, mean creatine kinase was significantly
reduced compared to baseline. All three participants showed
improvements from baseline across all functional measures.
No new safety signals were observed and the safety
profile seen to date supports the ability to dose escalate in the next
cohort of the study.
https://seekingalpha.com/news/3503968-sarepta-5-percent-premarket-positive-data-srpminus-9003-trialFDA approves AstraZeneca asthma drug for self-administration
AstraZeneca (NYSE:AZN) says the U.S. Food and Drug Administration approved the self-administration of its Fasenra asthma treatment, which uses a pre-filled, single-use auto-injector pen.
“We can now offer Fasenra in an even more
convenient way, giving U.S. healthcare providers and patients the option
of administering Fasenra at home or in a doctor’s office, and making
treatment more accessible to patients with severe eosinophilic asthma,”
the company says.
Fasenra, AZN’s first respiratory biologic, already
is approved as an add-on maintenance treatment in severe eosinophilic
asthma in the U.S., Europe and Japan; Fasenra self-administration and
the Fasenra Pen already are approved in the European Union.
https://seekingalpha.com/news/3503937-fda-approves-astrazeneca-asthma-drug-self-administrationSpectrum Pharma announces publication of positive poziotinib data in cancer cell
Spectrum Pharmaceuticals (NASDAQ:SPPI) announces
a publication entitled, “Pan-Cancer Landscape and Functional Analysis
of ERBB2 Mutations Identifies Poziotinib as a Clinically Active
Inhibitor and Enhancer of T-DM1 Activity.”
In Company’s pre-clinical study of 11 EGFR/HER2
tyrosine kinase inhibitors, poziotinib was the most potent HER2
mutant-selective TKI tested, and was found to be highly active in NSCLC
patients with HER2 exon 20 mutations.
Poziotinib is found to be synergistic with a
HER2-targeting antibody-drug conjugate, providing a rationale for
combinations to move forward into the clinic.
The Company plans to disclose topline data from
the first cohort of ZENITH20 study in current quarter. Topline data from
cohort 2 is expected by mid-2020.
Spectrum received exclusive license from Hanmi
Pharmaceuticals to develop and commercialize poziotinib worldwide,
excluding Korea and China.
Shares are up 2% premarket.
https://seekingalpha.com/news/3503957-spectrum-pharma-announces-publication-positive-poziotinib-data-cancer-cell
Value of Juul Labs stakes slashed
Hedge fund Darsana Capital reportedly wrote down its investment in Juul Labs (JUUL) by more than a third.
Sources says the Darsana stake in Juul is now valued at $24B after being valued at $38B a year ago.
Altria’s (NYSE:MO) in Juul stake is estimated to have dropped about 20% since its $12.8B investment. Shares of Altria are up 0.10% premarket to $40.85.
https://seekingalpha.com/news/3503961-value-juul-labs-stakes-slashedThursday, October 3, 2019
FDA Verdict Ahead For Pfenex’s Osteoporosis Biosimilar
Pfenex Inc PFNX 0.76%
has a key binary event coming up: mark your calendar for Oct. 7, when
the FDA is scheduled to issue its verdict on the company’s lead drug
candidate.
Through its production platform Pfenex Expression Technology, the biotech Pfenex engages in the development of high-value protein-based therapeutics and vaccines.
This thinly traded micro-cap biotech has more than doubled year-to-date. Has the rally run out of steam, or is there further upside ahead?
Through its production platform Pfenex Expression Technology, the biotech Pfenex engages in the development of high-value protein-based therapeutics and vaccines.
This thinly traded micro-cap biotech has more than doubled year-to-date. Has the rally run out of steam, or is there further upside ahead?
Clinical Pipeline
The most advanced product candidate in Pfenex’ pipeline, PF708, is inching closer to commercialization.Forteo’s global sales stood at $1.6 billion in 2019.
Following the midcycle review of Pfenex’s NDA for PF708 in May, the FDA did not identify any issues requiring an Adcom meeting, the company said in its second-quarter earnings release.
The company is gearing for a commercial launch in the U.S. as early as the fourth quarter of 2019, subject to FDA approval.
Pfenex is also striving to get an “A” therapeutic equivalence designation for PF708 to Forteo, which will allow PF708 to be automatically substituted for Forteo.
The company’s development and licensing partner Alvogen has assumed the responsibility to manufacture and commercialize PF708 in the U.S. at its own expense.
Alvogen has also been granted exclusive rights to market PF708 in the EU, certain countries in the Middle East and North Africa and the rest of the world, excluding those Asian countries for which China NT Pharma has secured the licensing rights.
Pfenex is eligible to receive a gross profit split of up to 60% on product sales from Alvogen, depending on the geography and cost of goods sold.
The company is eligible for double-digit royalties on net sales from NT Pharma.
Alvogen has submitted a Marketing Authorization Application to the European Medicines Agency. Initial comments on the application are expected in the third quarter of 2019.
The Pipeline
Pfenex’s other wholly owned investigational compounds include:-PF582 (randibizumab), a biosimilar candidate to Lucentis marketed by Roche Holdings AG Basel ADR RHHBY and Novartis AG NVS 1%, indicated to treat retinal diseases.
-PF529, a biosimilar candidate to Amgen, Inc. AMGN 1.8%‘s Neulasta for treating chemotherapy-induced neutropenia.
-PF690, a biosimilar product to Oncaspar, a drug indicated to treat acute lymphoblastic leukemia that’s marketed by Shire, which has been acquired by Takeda Pharmaceutical Co Ltd TAK 1.27%.
-Anthrax vaccine candidates Px563L/RPA563.
-Under partnered programs, Pfenex has PF743, a recombinant crisantaspase and PF745, a recombinant crisantaspase with a half-life extension technology, both produced using its PET platform under an agreement with Jazz Pharmaceuticals PLC JAZZ 1.56%.
PF743, which has been named JZP-458, has completed a Phase 1 study successfully, with Jazz aiming to start a Phase 2/3 study later this year.
 -Pfenex
has development and commercial partnerships for CRM-197, a non-toxic
mutant of diphtheria toxin, which functions as a carrier for
polysaccharides and haptens, making them immunogenic.
-Pfenex
has development and commercial partnerships for CRM-197, a non-toxic
mutant of diphtheria toxin, which functions as a carrier for
polysaccharides and haptens, making them immunogenic.It has commercial agreements in place with Merck & Co., Inc. MRK 1.33% and the Serum Institute of India, or SII.
Merck is using CRM197 in 12 Phase 3 studies for its V114, an investigational 15-valence conjugate vaccine for the prevention of pneumococcal disease.
SII has a 10-valence pneumococcal conjugate vaccine, Pneumosil, which utilizes CRM197. SII has initiated the process of World Health Organization prequalification for Pneumosil in the first quarter of 2019, with the process likely taking upto a year.
Competition
Apart from the $1.6 billion in sales netted by Eli Lilly, Radius Health Inc RDUS 2.56%‘s Tymlos, another approved osteoporosis drug, posted sales of $100 million, taking the total sales of osteoporosis drugs to $1.7 billion last year.Pfenex may also have to brace for additional competition, as Evenity developed by Amgen, Inc. AMGN 1.8% and Belgian biotech UCB has been given the nod in the U.S. for treating osteoporosis in menopausal women at a high risk of fracture, although the EU has yet to approve the drug.
Osteoporosis product sales are expected to reach $6.7 billion in the U.S., Japan and the five major European markets by 2025. About 53 million Americans either have osteoporosis or are at increased risk, Pfenex said, citing NIH estimates.
Financials
Revenue recognized by Pfenex is through collaboration agreements, licensing agreement, government contracts and sales of reagent protein products.A majority of the company’s 2018 revenues were derived from its collaboration agreement with Jazz and from its advanced development contract with BARDA, with each accounting for over 10% of its total revenues.
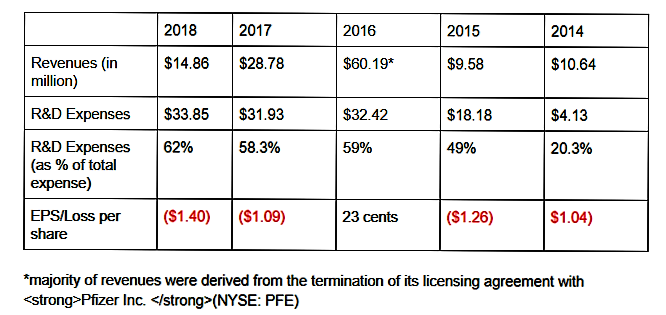 Source: 10-Q Filing
Source: 10-Q Filing For the three months ended June 30, 2019, the company reported revenue of $2.81 million, down from $4.19 million year-over-year.
The loss per share narrowed from 41 cents to 24 cents, as R&D expenses declined about 55%. Cash and cash equivalents stood at $41.62 million as of June 30.
Upcoming Catalysts
- Oct. 3: Presentation at the Cantor Healthcare Conference.
- Oct. 7: PDUFA Date For PF708.
- Commercial launch of PF708: as early as the fourth quarter of 2019 (contingent on FDA approval). Licensee Jazz to initiate Phase 2/3 study of PF743 (JZP-458): later this year.
- Potential sales royalty payments for the Serum Institute of India’s Pneumosal: as early as the first quarter of 2020, when the WHO prequalification process is complete.
Stock Take
Pfenex, which went public in July 2014 through an 8.33-million-share IPO priced at $6 per share, rose to a post-IPO high of $24.41 in a year.The stock then went downhill, troughing at $2.07 in December 2017 before attempting a modest recovery that took it to a peak of $8.42 in mid-2018.
Thereafter, it has been more of a consolidation move. The stock has traded in a 52-week range of $3.13-$10.20.

Source: Y Charts
From an near-term intraday low of $5.65 on Aug. 5, Pfenex shares rallied to a high of $10.20 on Sept. 17, a peak-trough gain of roughly 81%.
The catalyst for the recent rally has been the kickoff of a $17-million development milestone payment from Jazz for PF745.
The 50-day SMA has not been violated to the downside by the stock since early August, with the measure currently at $7.448.
Buying could emerge if the stock drops back to this level. The $7.20-$7.30 region served as resistance for the stock since the middle of May before buying pushed the stock past this resistance in early September.
If this resistance-turned support level breaks down, the next support could be around $5.40-$5.50, a low from which the stock has bounced a few times in recent months.
On the contrary, the stock could revisit its recent highs around $9.70-$10.20 on a potentially favorable reaction to the PDUFA catalyst.
Pfenex’s pipeline optionality and a steady stream of milestone payments from partnered programs and collaborations could cushion the stock even in the wake of an adverse outcome.
In early September, JMP Securities analyst Jason Butler upgraded shares of Pfenex from Market Perform to Outperform and raised the price target from $13 to $17, suggesting over 100% upside potential.
https://www.benzinga.com/general/biotech/19/10/14527152/biotech-stock-on-the-radar-fda-verdict-ahead-for-pfenexs-osteoporosis-biosimilar
MEI Pharma +16.7% on updated early-stage trial data
MEI Pharma (NASDAQ:MEIP) has risen another 16.7% postmarket after citing updated clinical data from an early-stage trial.
The company announced data from an ongoing Phase
1b study of investigational ME-401 in patients with indolent B-cell
malignancies.
It will present the data at the company’s investor and analyst event, starting tomorrow at 8 a.m. ET.
The company will also review progress across the
pipeline of its four clinical-stage oncology candidates, with a focus on
voruciclib, a cyclin-dependent kinase inhibitor with potent CDK9
inhibition.
https://seekingalpha.com/news/3503880-mei-pharma-plus-16_7-percent-updated-early-stage-trial-data
Subscribe to:
Posts (Atom)