Ohioans would be able to learn the bottom-line cost of their
scheduled hospital care — and their out-of-pocket share of the bill — in
advance under a bill passed unanimously by the Ohio Senate on
Wednesday.
Senate Bill 97,
sponsored by Sen. Steve Huffman, R-Tipp City, would require hospitals
to provide a “reasonable, good faith” cost estimate, or price range, of
scheduled services at the advance request of patients beginning July 1,
2021.
The information must include an estimate of out-of-pocket costs and
whether other costs, such as physician or anesthesiology services, will
be billed separately. Hospitals also must warn patients if they are out
of network with their health insurer to prevent so-called “surprise
billing.” Patients must request cost estimates at least seven days in
advance.
Hospitals also must provide a web site which contains the standard
costs of common items and services, which also is required under federal
law to allow consumers to price shop.
Senate President Larry Obhof, R-Medina, said the bill would give
consumers a heads-up on potential costs to avoid financial surprises and
could help control costs.
The bill, passed by a 32-0 vote, now advances to the House for consideration.
Lawmakers used the state budget to insert the hospital price
transparency language and a provision requiring insurers to cover
out-of-network care when provided at in-network facilities.
However, Republican Gov. Mike DeWine vetoed the provisions, calling
them unworkable and duplicative of federal requirements. A separate bill
addressing “surprise billing” accompanying out-of-network care at
covered providers is undergoing hearings in the Senate.
The price disclosure mandate comes as health care costs continue to
spiral. The Kaiser Family Foundation reports the average price of an
in-patient surgery increased 30% over five years. And, employee health
insurance premiums and deductibles for a family plan have increased 162%
over the last 10 years, the foundation reported.
The Ohio Hospital Association supports the bill, saying a
“competitive health care market and patient demand” has prompted
hospitals and other health care providers to publicly post costs for
common procedures and inform patients of their uninsured share of the
price.
“What patients want to know is how much something is going to cost
them out of their pocket,” Sean McGlone, lawyer for the hospital
association, told a Senate committee.
As introduced, the bill would have required physicians, therapists,
dentists, chiropractors, psychologists and others to also provide cost
estimates, but that portion was dropped due to concerns over burdensome
regulations on smaller practioners, which DeWine cited in his veto.
The General Assembly’s last attempt to improve cost transparency in
2015 died in the courts. After delaying its enactment, the Ohio Hospital
Association and other health care providers obtained a state court
injunction early this year, successfully arguing the bill violated the
“one bill, one topic” section of the Ohio Constitution by not being
passed as stand-alone legislation.
In other action, the Senate voted unanimously to approve Senate Bill 52,
creating the Ohio Cyber Reserve as civilian IT volunteers under the
Ohio National Guard to be called to duty in response to cyber attacks on
state and local government computer systems and other infrastructure.
To increase election security against hacking and other threats, the
bill makes Secretary of State Frank LaRose a member of Ohio’s Homeland
Security Advisory Council. The legislation also requires county boards
of election to audit the official results of general and primary
elections, which now occur under directives from the secretary of state.
The bill now goes to DeWine for his signature.
https://www.dispatch.com/news/20191009/senate-passes-bill-to-allow-patients-to-learn-hospital-costs-in-advance
Search This Blog
Wednesday, October 9, 2019
Return of Vioxx: Can drug once deemed deadly relaunch to treat rare disease?
Since its recall in 2004, the pain drug Vioxx has been a symbol of
pharmaceutical danger, starring in countless daytime legal
advertisements explaining how you, or perhaps a loved one, might be
entitled to millions in settlement dollars. But one company believes the
infamous drug deserves a second chance, and is plotting to resurrect
the former blockbuster as a treatment for a rare, incurable condition.
Vioxx’s days as a widely used treatment for arthritis and chronic pain came to an end when studies revealed that it roughly doubled patients’ risk of heart attack and stroke, leading to an estimated 60,000 deaths. It was a public health disaster, and it led to a congressional investigation, allegations of lapses at the Food and Drug Administration, and agreement by the manufacturer, Merck, to pay a nearly $5 billion settlement.
Now, Tremeau Pharmaceuticals, a private Massachusetts company, is developing a generic version of Vioxx as a treatment for severe joint pain in people with hemophilia, a side effect called hemophilic arthropathy. The company plans to begin a pivotal trial next year and, if the resulting data are positive, Tremeau will apply for FDA approval in hopes of bringing Vioxx back to market more than 15 years after Merck pulled it from shelves.
Experts say the drug’s cardiovascular issues are nothing to discount,
but Vioxx’s deadly history isn’t doomed to repeat itself. The alarming
number of heart attacks associated with the drug represent a small
percentage of the millions of people who took it, driven in part by the
fact that doctors prescribed liberally. Now that Vioxx’s relationship
with cardiovascular danger is well-understood, they say, doctors could
mitigate future problems by ensuring that patients at high risk don’t
get it, and by consistently monitoring those who do.
While the Vioxx brand might be tarnished in the popular consciousness, the drug has a much different reputation in the hemophilia community. Before its recall, the drug was the off-label drug of choice to treat hemophilic arthropathy, which results from a buildup of blood in patients’ joints and leads to pain, inflammation, and tissue damage. Over-the-counter pain treatments like ibuprofen can lead to internal bleeding, making them inadvisable for patients with hemophilia.
Dr. Ellis Neufeld, clinical director of hematology at St. Jude Children’s Research Hospital in Memphis, said the recall of Vioxx, while certainly advisable given the safety concerns, robbed patients with hemophilia of an important treatment.
“That was a bad day,” Neufeld said. “We really lamented it. Vioxx was a good drug.”
In the years since, doctors have reached for steroids, physical therapy, and even opioids to treat the pain of hemophilic arthropathy. In severe cases, patients get their problematic joints replaced. But each solution comes with limitations. Steroids can only be used for a short time, and opioids come with serious risks of abuse.
“Vioxx certainly left a void in our arsenal,” said Dr. Catherine Broome, a hematologist at Georgetown University Hospital in Washington, D.C.
While physicians who spoke with STAT have no trouble seeing the medical utility of a Vioxx reboot, the business case is a little less clear.
There are only about 20,000 people in the U.S. with hemophilia, and, thanks to modern treatment, the percentage who develop hemophilic arthropathy has decreased over time, doctors said. Since the 1990s, physicians have been able to replace the missing proteins that prevent clotting in patients with hemophilia, minimizing the number of bleeds and thus reducing the incidence of hemophilic arthropathy.
“If you take what we dealt with in the early years in my career, the people I was taking care of then, they are all severely affected with hemophilic arthropathy,” said Broome, who has been treating hemophilia patients since 1985. “But if you look at young men in their 20s and 30s right now, you can’t tell them apart from you or me.”
On the horizon are one-time gene therapies that promise to free hemophilia patients from regular infusions of clotting protein. The most advanced of those therapies, a treatment for hemophilia A developed by BioMarin, could win FDA approval as early as next year. If its effects prove to last a lifetime, the number of people with hemophilic arthropathy would further wane.
That could mean Tremeau’s reanimated Vioxx would be a niche product at best, and it might explain the company’s delayed development.
In 2017, Tremeau told the Associated Press that it would need to raise $25 million to fund its pivotal trial. That same year, the company said it “gained agreement” with the FDA on the size, duration, and details of that trial. However, Tremeau has raised just $5.2 million since then and is yet to enroll a single patient.
In an email, a Tremeau representative said the company is “in active discussions with the FDA and the hemophilia community” and plans to start the trial some time in 2020. The company declined to answer questions about the cause of its delay and whether it has enough money to complete the study.
Despite the changing dynamics in hemophilia treatment, “hemophilic arthropathy isn’t going away,” said Dr. Christopher Walsh, a hematologist at Mount Sinai Hospital in New York City who has served as a paid adviser to Tremeau.
“There’s a definite need in this community for alternative pain medication that is not addictive,” Walsh said. “And Vioxx — hopefully, if they do the study — would be one of those drugs. But that remains to be seen.”
Vioxx’s days as a widely used treatment for arthritis and chronic pain came to an end when studies revealed that it roughly doubled patients’ risk of heart attack and stroke, leading to an estimated 60,000 deaths. It was a public health disaster, and it led to a congressional investigation, allegations of lapses at the Food and Drug Administration, and agreement by the manufacturer, Merck, to pay a nearly $5 billion settlement.
Now, Tremeau Pharmaceuticals, a private Massachusetts company, is developing a generic version of Vioxx as a treatment for severe joint pain in people with hemophilia, a side effect called hemophilic arthropathy. The company plans to begin a pivotal trial next year and, if the resulting data are positive, Tremeau will apply for FDA approval in hopes of bringing Vioxx back to market more than 15 years after Merck pulled it from shelves.
While the Vioxx brand might be tarnished in the popular consciousness, the drug has a much different reputation in the hemophilia community. Before its recall, the drug was the off-label drug of choice to treat hemophilic arthropathy, which results from a buildup of blood in patients’ joints and leads to pain, inflammation, and tissue damage. Over-the-counter pain treatments like ibuprofen can lead to internal bleeding, making them inadvisable for patients with hemophilia.
Dr. Ellis Neufeld, clinical director of hematology at St. Jude Children’s Research Hospital in Memphis, said the recall of Vioxx, while certainly advisable given the safety concerns, robbed patients with hemophilia of an important treatment.
“That was a bad day,” Neufeld said. “We really lamented it. Vioxx was a good drug.”
In the years since, doctors have reached for steroids, physical therapy, and even opioids to treat the pain of hemophilic arthropathy. In severe cases, patients get their problematic joints replaced. But each solution comes with limitations. Steroids can only be used for a short time, and opioids come with serious risks of abuse.
“Vioxx certainly left a void in our arsenal,” said Dr. Catherine Broome, a hematologist at Georgetown University Hospital in Washington, D.C.
While physicians who spoke with STAT have no trouble seeing the medical utility of a Vioxx reboot, the business case is a little less clear.
There are only about 20,000 people in the U.S. with hemophilia, and, thanks to modern treatment, the percentage who develop hemophilic arthropathy has decreased over time, doctors said. Since the 1990s, physicians have been able to replace the missing proteins that prevent clotting in patients with hemophilia, minimizing the number of bleeds and thus reducing the incidence of hemophilic arthropathy.
“If you take what we dealt with in the early years in my career, the people I was taking care of then, they are all severely affected with hemophilic arthropathy,” said Broome, who has been treating hemophilia patients since 1985. “But if you look at young men in their 20s and 30s right now, you can’t tell them apart from you or me.”
On the horizon are one-time gene therapies that promise to free hemophilia patients from regular infusions of clotting protein. The most advanced of those therapies, a treatment for hemophilia A developed by BioMarin, could win FDA approval as early as next year. If its effects prove to last a lifetime, the number of people with hemophilic arthropathy would further wane.
That could mean Tremeau’s reanimated Vioxx would be a niche product at best, and it might explain the company’s delayed development.
In 2017, Tremeau told the Associated Press that it would need to raise $25 million to fund its pivotal trial. That same year, the company said it “gained agreement” with the FDA on the size, duration, and details of that trial. However, Tremeau has raised just $5.2 million since then and is yet to enroll a single patient.
In an email, a Tremeau representative said the company is “in active discussions with the FDA and the hemophilia community” and plans to start the trial some time in 2020. The company declined to answer questions about the cause of its delay and whether it has enough money to complete the study.
Despite the changing dynamics in hemophilia treatment, “hemophilic arthropathy isn’t going away,” said Dr. Christopher Walsh, a hematologist at Mount Sinai Hospital in New York City who has served as a paid adviser to Tremeau.
“There’s a definite need in this community for alternative pain medication that is not addictive,” Walsh said. “And Vioxx — hopefully, if they do the study — would be one of those drugs. But that remains to be seen.”
The return of Vioxx: Can a drug once deemed deadly be relaunched to treat rare disease?
Alibaba to stop sales of e-cigarette components in US
Chinese e-commerce firm Alibaba said on Wednesday it will stop
selling e-cigarette components in the United States, amid growing
regulatory scrutiny and reports of lung disease and some deaths linked
to vaping.
The move follows announcements by Kroger Co and Walgreens Boots Alliance Inc this week that they would stop selling e-cigarettes at their stores, in line with a similar decision by Walmart.
Alibaba said it already had a long-standing policy in place to not sell complete e-cigarette products in the United States.
Vaping products have been linked to a mysterious lung illness that is
reported to have led to 18 deaths as of last week, with the number of
confirmed and probable cases of the condition exceeding 1,000, according
to the U.S. Centers for Disease Control and Prevention.
Alibaba Group Holding Ltd said that listings for products such as box mods, vape pens, herbal vapors, heat not burn devices, and empty pod cartridges would not be displayed for users located in the United States.
While Juul Labs Inc dominates the North American market for pod e-cigarettes, many reports of death and injury in the United States have been tied to makeshift brands with no identifiable owner.
The most prominent, Dank Vapes, was linked to 24 patients with lung
illness, according to a study from the New England Journal of Medicine.
The products contained THC, the psychoactive ingredient in marijuana.
Prior to the suspension, buyers could easily purchase devices, component parts and packaging from sites like Alibaba or Amazon to make their own counterfeit vaping devices.
Amazon.com Inc took down vape paraphernalia in September, although it did not specify the exact products it removed.
https://www.reuters.com/article/us-alibaba-cigarettes/alibaba-to-stop-sales-of-e-cigarette-components-in-united-states-idUSKBN1WO0A3
The move follows announcements by Kroger Co and Walgreens Boots Alliance Inc this week that they would stop selling e-cigarettes at their stores, in line with a similar decision by Walmart.
Alibaba said it already had a long-standing policy in place to not sell complete e-cigarette products in the United States.
Alibaba Group Holding Ltd said that listings for products such as box mods, vape pens, herbal vapors, heat not burn devices, and empty pod cartridges would not be displayed for users located in the United States.
While Juul Labs Inc dominates the North American market for pod e-cigarettes, many reports of death and injury in the United States have been tied to makeshift brands with no identifiable owner.
Prior to the suspension, buyers could easily purchase devices, component parts and packaging from sites like Alibaba or Amazon to make their own counterfeit vaping devices.
Amazon.com Inc took down vape paraphernalia in September, although it did not specify the exact products it removed.
https://www.reuters.com/article/us-alibaba-cigarettes/alibaba-to-stop-sales-of-e-cigarette-components-in-united-states-idUSKBN1WO0A3
Administration to change anti-kickback rules for healthcare providers
The Trump administration will announce plans to change healthcare
regulations on Wednesday to loosen anti-kickback provisions that
restrict the kinds of outside services providers can refer patients to,
administration officials said.
President Donald Trump on Thursday will explain how the new rules advance his broader healthcare agenda, which includes reducing regulatory burdens and promoting innovative ways to reimburse healthcare providers, in a speech in Minnesota, the officials said.
The plan will change how the Department of Health and Human Services (HHS) enforces the Physician Self Referral Law, also known as the Stark law, which penalizes healthcare providers for referring patients to outside services that the provider could stand to benefit from financially.
HHS will create exceptions for healthcare providers that enter into
agreements with other parties if they are aimed at cutting costs and
improving patient health, the officials said.
Trump issued an executive order last week that sought to woo seniors by strengthening the Medicare health program.
The order was the Republican president’s answer to Democrats like Bernie Sanders, who is running to become the party’s nominee in the 2020 presidential election and is promoting the idea of Medicare for all Americans.
The Trump administration has also rolled out measures in recent
months designed to curtail drug prices and address other problems in the
U.S. healthcare system.
Policy experts say the efforts are unlikely to slow the rise of drug prices in a meaningful way, however.
https://www.reuters.com/article/us-usa-healthcare-trump/trump-administration-to-announce-changes-to-anti-kickback-rules-for-healthcare-providers-idUSKBN1WO1FP
President Donald Trump on Thursday will explain how the new rules advance his broader healthcare agenda, which includes reducing regulatory burdens and promoting innovative ways to reimburse healthcare providers, in a speech in Minnesota, the officials said.
The plan will change how the Department of Health and Human Services (HHS) enforces the Physician Self Referral Law, also known as the Stark law, which penalizes healthcare providers for referring patients to outside services that the provider could stand to benefit from financially.
Trump issued an executive order last week that sought to woo seniors by strengthening the Medicare health program.
The order was the Republican president’s answer to Democrats like Bernie Sanders, who is running to become the party’s nominee in the 2020 presidential election and is promoting the idea of Medicare for all Americans.
Policy experts say the efforts are unlikely to slow the rise of drug prices in a meaningful way, however.
https://www.reuters.com/article/us-usa-healthcare-trump/trump-administration-to-announce-changes-to-anti-kickback-rules-for-healthcare-providers-idUSKBN1WO1FP
NYC sues online e-cigarette retailers over age verification
New York City has sued more nearly two dozen online e-cigarette
retailers, accusing them of selling their products to underage New
Yorkers, the city announced Wednesday.
The lawsuit, filed Tuesday evening in Brooklyn federal court, targets
22 companies including Artison Vapor Franchise LLC, Eliquidstop.com and
Vapor 4 Life Holdings Inc. All of them are located outside New York.
The lawsuit accuses the companies of violating New York City law prohibiting the sale of e-cigarettes to people under 21 by marketing to underage consumers and failing to take adequate steps to verify their ages.
It seeks an order requiring them to comply with the law, and money damages “to compensate the city for the costs of abating the epidemic of underage e-cigarette use in the city.”
“The kids of New York are the pride of our city, but to these
companies, they’re just a source of profit,” New York City Mayor Bill de
Blasio said in a statement. “Preying on minors and hooking them on a
potentially lethal, lifelong nicotine addiction is unconscionable.”
Artisan Vapor Franchise, Eliquidstop.com and Vapor 4 Life could not immediately be reached for comment.
There has been a regulatory crackdown on e-cigarettes and a growing number of lawsuits by young adults and parents of teenagers against leading e-cigarette company Juul Labs Inc and its parent company, Marlboro maker Altria Group.
Most of the lawsuits say only that users became addicted to nicotine as a result of using Juul, but some allege serious health consequences.
Some states, including New York, Michigan and Rhode Island, have moved to restrict the sale of flavored e-cigarettes. Massachusetts has gone further, instituting a four-month ban on all vaping products.
Last week, a New York judge blocked a state ban on most flavored e-cigarettes from taking effect after an industry group sued to challenge it, while a Massachusetts judge upheld that state’s ban.
https://www.reuters.com/article/us-new-york-vaping-lawsuit/new-york-city-sues-online-e-cigarette-retailers-over-age-verification-idUSKBN1WO2IG
The lawsuit accuses the companies of violating New York City law prohibiting the sale of e-cigarettes to people under 21 by marketing to underage consumers and failing to take adequate steps to verify their ages.
It seeks an order requiring them to comply with the law, and money damages “to compensate the city for the costs of abating the epidemic of underage e-cigarette use in the city.”
Artisan Vapor Franchise, Eliquidstop.com and Vapor 4 Life could not immediately be reached for comment.
There has been a regulatory crackdown on e-cigarettes and a growing number of lawsuits by young adults and parents of teenagers against leading e-cigarette company Juul Labs Inc and its parent company, Marlboro maker Altria Group.
Most of the lawsuits say only that users became addicted to nicotine as a result of using Juul, but some allege serious health consequences.
Some states, including New York, Michigan and Rhode Island, have moved to restrict the sale of flavored e-cigarettes. Massachusetts has gone further, instituting a four-month ban on all vaping products.
Last week, a New York judge blocked a state ban on most flavored e-cigarettes from taking effect after an industry group sued to challenge it, while a Massachusetts judge upheld that state’s ban.
https://www.reuters.com/article/us-new-york-vaping-lawsuit/new-york-city-sues-online-e-cigarette-retailers-over-age-verification-idUSKBN1WO2IG
Humans have salamander-like ability to regrow cartilage in joints
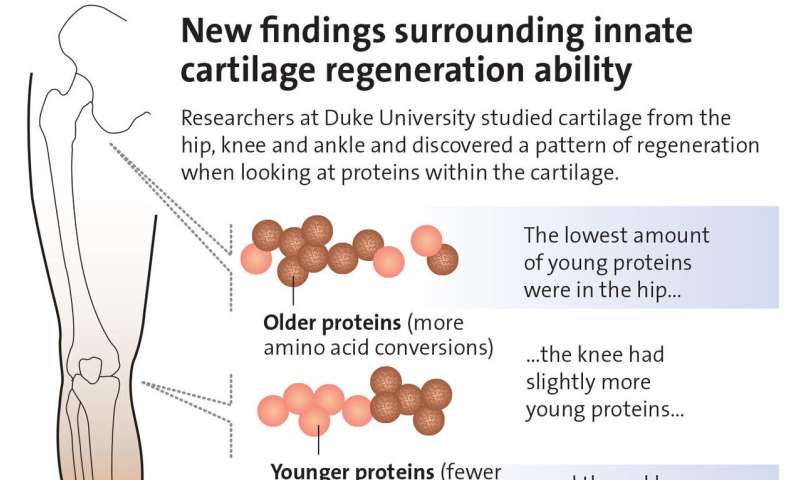
“We believe that an understanding of this ‘salamander-like’ regenerative capacity in humans, and the critically missing components of this regulatory circuit, could provide the foundation for new approaches to repair joint tissues and possibly whole human limbs,” said senior author Virginia Byers Kraus, M.D., Ph.D., a professor in the departments of Medicine, Pathology and Orthopedic Surgery at Duke.
Kraus and colleagues, including lead author Ming-Feng Hsueh, Ph.D., devised a way to determine the age of proteins using internal molecular clocks integral to amino acids, which convert one form to another with predictable regularity.
Newly created proteins in tissue have few or no amino acid conversions; older proteins have many. Understanding this process enabled the researchers to use sensitive mass spectrometry to identify when key proteins in human cartilage, including collagens, were young, middle-aged or old.
They found that the age of cartilage largely depended on where it resided in the body. Cartilage in ankles is young, it’s middle-aged in the knee and old in the hips. This correlation between the age of human cartilage and its location in the body aligns with how limb repair occurs in certain animals, which more readily regenerate at the furthest tips, including the ends of legs or tails.
The finding also helps explain why injuries to people’s knees and, especially, hips take a long time to recover and often develop into arthritis, while ankle injuries heal quicker and less often become severely arthritic.
The researchers further learned that molecules called microRNA regulate this process. Not surprisingly, these microRNAs are more active in animals that are known for limb, fin or tail repair, including salamanders, zebrafish, African fresh water fish and lizards.
These microRNAs are also found in humans—an evolutionary artifact that provides the capability in humans for joint tissue repair. As in animals, microRNA activity varies significantly by its location: it was highest in ankles compared to knees and hips and higher in the top layer of cartilage compared to deeper layers of cartilage.
“We were excited to learn that the regulators of regeneration in the salamander limb appear to also be the controllers of joint tissue repair in the human limb,” Hsueh said. “We call it our ‘inner salamander’ capacity.”
The researchers said microRNAs could be developed as medicines that might prevent, slow or reverse arthritis.
“We believe we could boost these regulators to fully regenerate degenerated cartilage of an arthritic joint. If we can figure out what regulators we are missing compared with salamanders, we might even be able to add the missing components back and develop a way someday to regenerate part or all of an injured human limb,” Kraus said. “We believe this is a fundamental mechanism of repair that could be applied to many tissues, not just cartilage.”
Explore further
More information: “Analysis of “old” proteins unmasks dynamic gradient of cartilage turnover in human limbs” Science Advances (2019). advances.sciencemag.org/content/5/10/eaax3203
FDA classifies Medtronic catheter recall as Class I
Medtronic’s (NYSE:MDT) voluntary recall
of its 6F Sherpa NX Active Coronary Guide Catheter is considered Class I
by the FDA, its highest level since the product issue could cause
serious injury or death.
Affected catheters have a risk of losing outer
material from a distal component which could result in the underlying
stainless steel braid wires being exposed after insertion, although the
company says it has not received any reports of patient injuries to
date. The company investigated the issue after receiving six complaints.
https://seekingalpha.com/news/3504832-fda-classifies-medtronic-catheter-recall-class
Subscribe to:
Posts (Atom)