In a 14-2 vote, the FDA’s Antimicrobial Drugs Advisory Committee backed Shionogi’s (OTCPK:SGIOY) cefiderocol for the treatment of complicated urinary tract infection.
https://seekingalpha.com/news/3506356-fda-ad-com-thumbs-shionogis-cefiderocol
Search This Blog
Wednesday, October 16, 2019
After-hours analyst action, Oct. 16
Moleculin Biotech (NASDAQ:MBRX) resumed with Outperform rating and $2 (94% upside) price target at Oppenheimer. Shares up 4% after hours.
Vertex Pharmaceuticals (NASDAQ:VRTX) resumed with Buy rating and $220 (25% upside) price target at BofA Merrill Lynch.
Regeneron Pharmaceuticals (NASDAQ:REGN) resumed with Neutral rating and $325 (9% upside) price target at BofAML.
Merck (NYSE:MRK) resumed with Neutral rating and $90 (7% upside) price target at BofAML.
Pfizer (NYSE:PFE) resumed with Neutral rating and $37 (2% upside) price target at BofAML.
Eli Lilly (NYSE:LLY) resumed with a Buy rating and $133 (23% upside) price target at BofAML.
Gilead Sciences (NASDAQ:GILD) resumed with Neutral rating and $70 (7% upside) price target at BofAML.
Bristol-Myers Squibb (NYSE:BMY) resumed with Buy rating and $60 (16% upside) price target at BofAML.
BioMarin Pharmaceutical (NASDAQ:BMRN) resumed with Buy rating and $90 (32% upside) price target at BofAML. Shares up 1% after hours.
Amgen (NASDAQ:AMGN) resumed with Neutral rating and $215 (6% upside) price target at BofAML.
Biogen (NASDAQ:BIIB) resumed with Underperform rating and $200 (12% downside risk) price target at BofAML. Shares down a fraction after hours.
https://seekingalpha.com/news/3506336-bofa-merrill-lynch-sees-25-percent-upside-vertex-hours-analyst-actionGilead chief strategist gets big promotion after orchestrating multi-$B deals
After gaining credit as the architect of Gilead’s $12 billion Kite
buyout as well as the recent $5 billion partnership with Galapagos,
chief strategy officer Andrew Dickinson is being promoted to the
prestigious CFO post at the big biotech. And new CEO Daniel O’Day says
the latest move completes his makeover of the top team.
Dickinson will remain in charge of strategy in his new post.
A 3-year veteran at Gilead, Dickinson joined the bellwether biotech after a lengthy stint at Lazard Frères & Co, where he was global co-head of healthcare investing. Before that, ironically enough, he had been at Myogen, which was bought out by Gilead in 2006. Now he’ll be primarily responsible for building confidence in the numbers at a company that has a strong foundation in HIV, a disappearing franchise in hep C and a CAR-T subsidiary in Kite that has a long way to go in establishing a new business.
Dickinson will get some guidance from Robin Washington, who he will be replacing as CFO. Washington said earlier that she would remain as an adviser for the company as she exited the C-suite.
O’Day has been making some sweeping changes at the top end of the company since taking the helm. Partly that was due to an exodus of execs out of R&D as they moved to new jobs running their own biotech companies. And CEO John Milligan and chairman John Martin both left in tandem as Gilead sought a new direction in drug development that would please an anxious Wall Street.
That’s a work in progress. And Jefferies’ Michael Yee counseled patience to investors anxious to see bottom-line improvements.
In the last 6 months, O’Day (1) filled the Kite CEO role from LLY, (2) put in a new head of Commercial from BMY, (3) hired a CMO role from Genentech, and (4) filled the CFO role internally as Dickinson was EVP Corporate Development/Strategy already and has been at GILD since 2016. Of note, Dickinson was brought on to join GILD during the later part of the John Milligan era. Dickinson was close with Milligan previously having served for a decade at Lazard to help advise on strategic ideas including BD and M&A. Bottom line for GILD – investors will need patience. Improved stories aren’t made overnight.
“Andy is an exceptional, highly strategic leader. In addition to his impressive business and financial acumen and broad experience, Andy possesses strong creativity and vision. This has been evident in the way Gilead has approached acquisitions and partnerships under Andy’s leadership,” noted O’Day. “Over the past months, one of my key priorities has been to ensure we have an outstanding team of leaders to shape Gilead’s long-term success. I am pleased that with Andy’s appointment as CFO, we now have our full leadership team in place.”
Dickinson will remain in charge of strategy in his new post.
A 3-year veteran at Gilead, Dickinson joined the bellwether biotech after a lengthy stint at Lazard Frères & Co, where he was global co-head of healthcare investing. Before that, ironically enough, he had been at Myogen, which was bought out by Gilead in 2006. Now he’ll be primarily responsible for building confidence in the numbers at a company that has a strong foundation in HIV, a disappearing franchise in hep C and a CAR-T subsidiary in Kite that has a long way to go in establishing a new business.
Dickinson will get some guidance from Robin Washington, who he will be replacing as CFO. Washington said earlier that she would remain as an adviser for the company as she exited the C-suite.
O’Day has been making some sweeping changes at the top end of the company since taking the helm. Partly that was due to an exodus of execs out of R&D as they moved to new jobs running their own biotech companies. And CEO John Milligan and chairman John Martin both left in tandem as Gilead sought a new direction in drug development that would please an anxious Wall Street.
That’s a work in progress. And Jefferies’ Michael Yee counseled patience to investors anxious to see bottom-line improvements.
In the last 6 months, O’Day (1) filled the Kite CEO role from LLY, (2) put in a new head of Commercial from BMY, (3) hired a CMO role from Genentech, and (4) filled the CFO role internally as Dickinson was EVP Corporate Development/Strategy already and has been at GILD since 2016. Of note, Dickinson was brought on to join GILD during the later part of the John Milligan era. Dickinson was close with Milligan previously having served for a decade at Lazard to help advise on strategic ideas including BD and M&A. Bottom line for GILD – investors will need patience. Improved stories aren’t made overnight.
“Andy is an exceptional, highly strategic leader. In addition to his impressive business and financial acumen and broad experience, Andy possesses strong creativity and vision. This has been evident in the way Gilead has approached acquisitions and partnerships under Andy’s leadership,” noted O’Day. “Over the past months, one of my key priorities has been to ensure we have an outstanding team of leaders to shape Gilead’s long-term success. I am pleased that with Andy’s appointment as CFO, we now have our full leadership team in place.”
Gilead’s chief strategy exec gets a big promotion after orchestrating multibillion-dollar deals
With new leadership, Sandoz breaks up with digital therapeutics outfit Pear
Looks like Novartis’ Sandoz unit — under new leadership — has severed
ties with Pear Therapeutics, dissolving the partnership that was inked
in 2018.
Last year, Sandoz and Pear collaborated on and launched reSET, an FDA-approved digital therapeutic designed to deliver cognitive behavioral therapy over 12 weeks to patients with substance abuse disorder (SUD) who are in outpatient treatment under the supervision of a doctor. The Swiss drugmaker also invested in Pear’s Series B round of financing announced in January 2018.
The future of Novartis’ generics unit Sandoz is uncertain — as price erosion in the United States fuels speculation that Novartis chief Vas Narasimhan might prefer to spin it off to sharpen focus on developing new treatments.
In March, Sandoz CEO Richard Francis abruptly tendered his resignation, suggesting he couldn’t commit to the ‘multi-year’ transformation that Sandoz was embarking on. Francis was replaced by Richard Saynor, SVP classic & established products, commercial & digital platforms at GSK — although he was with Sandoz prior to that stint.
The “decision to transition commercialization responsibilities for reSET and reSET-O is part of Sandoz transformation and subsequent leadership change, which has resulted in a reinforced focus on and capital allocation for Sandoz core business,” the company said in a statement on Tuesday.
Prescription digital therapeutics — such as reSET and Abilify Mycite, the first digital pill that carries an embedded sensor to track if patients are taking their medication properly — are validated in randomized clinical trials to demonstrate safety and efficacy.
The FDA approved reSET in 2017, on the basis of a NIDA-sponsored trial involving 399 patients with SUD. Patients were randomized to receive standard treatment — comprising intensive face-to-face counseling — or reduced amount of face-to-face counseling plus the digital therapeutic. Patients on the digital therapeutic more than doubled the rate of abstinence compared to standard face-to-face counseling. Pear Therapeutics is also developing digital therapeutics for a host of other disorders including schizophrenia, PTSD and general anxiety disorder. In December 2018, the FDA approved reSET-O, for the opioid use disorder.
Digital therapeutics is an umbrella term that includes technology such as wearable devices, mobile apps and telemedicine platforms — which is typically driven by software to prevent, manage, or treat disorders, independently or in concert with medication and/or medical devices. These tools are largely designed to address chronic diseases such as diabetes, heart or respiratory disorders, by targeting behaviors such as diet, exercise, and lifestyle that have a significant impact on the incidence and management of disease.
Although the rapid penetration of smartphones and tablets and lower healthcare costs have driven the growth of the global digital therapeutics market, privacy concerns could temper the pace of adoption. Still, the size of the global digital therapeutics market is expected to hit about $7.83 billion by 2025, from $1.75 billion in 2017 — according to Allied Market Research estimates.
Last year, Sandoz and Pear collaborated on and launched reSET, an FDA-approved digital therapeutic designed to deliver cognitive behavioral therapy over 12 weeks to patients with substance abuse disorder (SUD) who are in outpatient treatment under the supervision of a doctor. The Swiss drugmaker also invested in Pear’s Series B round of financing announced in January 2018.
The future of Novartis’ generics unit Sandoz is uncertain — as price erosion in the United States fuels speculation that Novartis chief Vas Narasimhan might prefer to spin it off to sharpen focus on developing new treatments.
In March, Sandoz CEO Richard Francis abruptly tendered his resignation, suggesting he couldn’t commit to the ‘multi-year’ transformation that Sandoz was embarking on. Francis was replaced by Richard Saynor, SVP classic & established products, commercial & digital platforms at GSK — although he was with Sandoz prior to that stint.
The “decision to transition commercialization responsibilities for reSET and reSET-O is part of Sandoz transformation and subsequent leadership change, which has resulted in a reinforced focus on and capital allocation for Sandoz core business,” the company said in a statement on Tuesday.
Prescription digital therapeutics — such as reSET and Abilify Mycite, the first digital pill that carries an embedded sensor to track if patients are taking their medication properly — are validated in randomized clinical trials to demonstrate safety and efficacy.
The FDA approved reSET in 2017, on the basis of a NIDA-sponsored trial involving 399 patients with SUD. Patients were randomized to receive standard treatment — comprising intensive face-to-face counseling — or reduced amount of face-to-face counseling plus the digital therapeutic. Patients on the digital therapeutic more than doubled the rate of abstinence compared to standard face-to-face counseling. Pear Therapeutics is also developing digital therapeutics for a host of other disorders including schizophrenia, PTSD and general anxiety disorder. In December 2018, the FDA approved reSET-O, for the opioid use disorder.
Digital therapeutics is an umbrella term that includes technology such as wearable devices, mobile apps and telemedicine platforms — which is typically driven by software to prevent, manage, or treat disorders, independently or in concert with medication and/or medical devices. These tools are largely designed to address chronic diseases such as diabetes, heart or respiratory disorders, by targeting behaviors such as diet, exercise, and lifestyle that have a significant impact on the incidence and management of disease.
Although the rapid penetration of smartphones and tablets and lower healthcare costs have driven the growth of the global digital therapeutics market, privacy concerns could temper the pace of adoption. Still, the size of the global digital therapeutics market is expected to hit about $7.83 billion by 2025, from $1.75 billion in 2017 — according to Allied Market Research estimates.
Under new leadership, Novartis’ Sandoz unit breaks up with digital therapeutics outfit Pear
Biosimilar threat under control for now, Roche savors demand for its new drugs
Roche $RHBBY unveiled a cheerful update on its performance in the
first nine months of this year, with demand for its newer drugs —
primarily in oncology/immunology — driving sales, particularly in China
where cancer is on the rise. In its press release, the Swiss drugmaker
mentioned its pending $4.3 billion acquisition of Spark Therapeutics
$ONCE, promising the deal — which has seen delay after delay due to
unrelenting scrutiny by competition regulators — will be consummated by
the end of the year.
The company — the world’s largest cancer drugmaker — lifted its full-year sales forecast, spurred by a 12% jump to about $36.7 billion in pharmaceutical sales. Sales were fueled by demand for Roche’s multiple sclerosis drug Ocrevus, hemophilia treatment Hemlibra, and its oncology products: checkpoint inhibitor Tecentriq and monoclonal antibody Perjeta and older Avastin — which more than offset declines in the sales of its cancer drugs Herceptin and MabThera/Rituxan.
Sales in Europe were down 1% due to Herceptin (-44%) and MabThera/Rituxan (-33%), but the strong growth of newer treatments almost steadied the ship.
“The impact of biosimilars (in Europe) is tailing off,” noted Roche’s head of pharmaceuticals Bill Anderson in a post-earnings conference call on Wednesday. Herceptin biosimilars launched in Europe about two years ago, while MabThera biosimilar erosion in the region kicked off last year.
Roche’s three legacy oncology drugs — Rituxan, Herceptin, and Avastin — are ripe for biosimilar competition in the United States — and earlier this year the company saw the launch of the first Herceptin and Avastin biosimilar. (Although if other biosimilar launches are any kind of barometer, the erosion of drug franchises in the United States is fairly muted compared to Europe.)
Still, Anderson preached caution. “We do expect there to be significant impact from biosimilars in the US, because there will be biosimilars to all three of our legacy oncology products — but also because there’s a need for additional competition, and we’ve been expecting it.”
He predicted the first five biosimilars for Rituxan will come sometime over the next quarter, with at least a couple of more Herceptin biosimilars in the next six months, and another Avastin biosimilar at the end of the year and/or early 2020.
International sales grew a healthy 20% mainly due to a strong uptake in China, where rates of cancer are high due to pollution and smoking. At some point the company will see limited biosimilar competition in China, although “there may be some questions about the quality of some of those early biosimilars so I think will we see ourselves as able to compete,” Anderson said. “In fact, one of the ways that will be competing in China is getting drugs like Perjeta and Hemlibra added to the national drug reimbursement list.”
As is the tradition, the company also culled a number of experimental drugs across its pipeline:
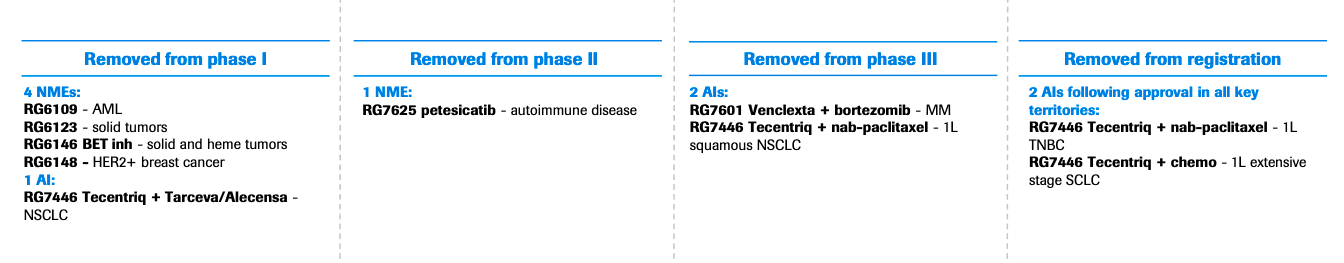
Source: Roche, 2019
One of the key cuts is petesicatib (RG7625), which was being developed to treat coeliac disease and autoimmune disorders, including Sjögren’s syndrome and psoriasis. The oral compound was engineered to target cathepsin-S. Years ago, Merck had touted the commercial potential of its cathepsin K inhibitor for osteoporosis, although it was forced to terminate the program after it was linked to an increased risk of cardiovascular events.
At the tail end of the conference call, the question of the Spark acquisition finally cropped up. Roche chief Severin Schwan did not go into specifics but reiterated that he was confident the deal would be completed by year-end.
The company — the world’s largest cancer drugmaker — lifted its full-year sales forecast, spurred by a 12% jump to about $36.7 billion in pharmaceutical sales. Sales were fueled by demand for Roche’s multiple sclerosis drug Ocrevus, hemophilia treatment Hemlibra, and its oncology products: checkpoint inhibitor Tecentriq and monoclonal antibody Perjeta and older Avastin — which more than offset declines in the sales of its cancer drugs Herceptin and MabThera/Rituxan.
Sales in Europe were down 1% due to Herceptin (-44%) and MabThera/Rituxan (-33%), but the strong growth of newer treatments almost steadied the ship.
“The impact of biosimilars (in Europe) is tailing off,” noted Roche’s head of pharmaceuticals Bill Anderson in a post-earnings conference call on Wednesday. Herceptin biosimilars launched in Europe about two years ago, while MabThera biosimilar erosion in the region kicked off last year.
Roche’s three legacy oncology drugs — Rituxan, Herceptin, and Avastin — are ripe for biosimilar competition in the United States — and earlier this year the company saw the launch of the first Herceptin and Avastin biosimilar. (Although if other biosimilar launches are any kind of barometer, the erosion of drug franchises in the United States is fairly muted compared to Europe.)
Still, Anderson preached caution. “We do expect there to be significant impact from biosimilars in the US, because there will be biosimilars to all three of our legacy oncology products — but also because there’s a need for additional competition, and we’ve been expecting it.”
He predicted the first five biosimilars for Rituxan will come sometime over the next quarter, with at least a couple of more Herceptin biosimilars in the next six months, and another Avastin biosimilar at the end of the year and/or early 2020.
International sales grew a healthy 20% mainly due to a strong uptake in China, where rates of cancer are high due to pollution and smoking. At some point the company will see limited biosimilar competition in China, although “there may be some questions about the quality of some of those early biosimilars so I think will we see ourselves as able to compete,” Anderson said. “In fact, one of the ways that will be competing in China is getting drugs like Perjeta and Hemlibra added to the national drug reimbursement list.”
As is the tradition, the company also culled a number of experimental drugs across its pipeline:

Source: Roche, 2019
One of the key cuts is petesicatib (RG7625), which was being developed to treat coeliac disease and autoimmune disorders, including Sjögren’s syndrome and psoriasis. The oral compound was engineered to target cathepsin-S. Years ago, Merck had touted the commercial potential of its cathepsin K inhibitor for osteoporosis, although it was forced to terminate the program after it was linked to an increased risk of cardiovascular events.
At the tail end of the conference call, the question of the Spark acquisition finally cropped up. Roche chief Severin Schwan did not go into specifics but reiterated that he was confident the deal would be completed by year-end.
With biosimilar threat under control (for now), Roche savors demand for its new drugs
Diplomat Pharmacy divests certain non-core assets to Diligent Health Solutions
Diplomat Pharmacy (NYSE:DPLO) has completed the sale of certain assets of Envoy Health Management, LLC to Diligent Health Solutions, LLC for an undisclosed sum.
Contracts administered at Envoy’s Chantilly, VA site and associated employees were included in the transaction.
https://seekingalpha.com/news/3506342-diplomat-pharmacy-divests-certain-non-core-assets-diligent-health-solutionsJ&J Offers $4 Billion Opioid Litigation Settlement
Johnson & Johnson has offered to pay about $4 billion to settle
all lawsuits in the U.S. accusing the company of contributing to the
opioid-addiction epidemic, according to people familiar with the matter.
If completed, the deal would resolve more than 2,000 lawsuits by state and local governments alleging that J&J’s marketing of pain drugs including Duragesic and Nucynta fueled the opioid crisis. J&J sold the U.S. rights to Nucynta to another company in 2015. The suits also allege that J&J is culpable for opioid abuse because of former divisions, which the company sold in 2016, that supplied active ingredients for other opioid manufacturers.
Bloomberg News reported the amount of J&J’s settlement offer Tuesday. The Wall Street Journal reported Tuesday that three major drug distributors were in talks to pay $18 billion to settle the opioid litigation, and that J&J was involved in the discussions to contribute additional money.
Teva Pharmaceutical Industries Inc. also is in talks to settle the opioid lawsuits against it by providing free drugs, according to people familiar with the matter.
A $4 billion settlement would probably please J&J investors, because analysts had expected the company would have to pay more. Wells Fargo had estimated that the opioid litigation could cost the company $5 billion to $10 billion to settle. Wall Street’s concerns about the litigation’s cost have shadowed J&J stock.
In August, an Oklahoma judge ordered J&J to pay $572 million for its role in the state’s opioid crisis, following a trial that began in May. On Tuesday, the judge said he made a miscalculation that overstated the award by more than $100 million, which he said will be reflected in his final order.
J&J said in August it would appeal the judgment and that the judge’s conclusions disregard the company’s compliance with federal and state laws.
The New Brunswick, N.J., company also recently agreed to a $20.4 million settlement of opioid lawsuits brought by two Ohio counties, avoiding a trial in the case scheduled to start next week.
J&J Chief Financial Officer Joseph Wolk told analysts on a conference call Tuesday regarding quarterly financial results that the company was open to settlements of opioid lawsuits like the ones in Ohio.
https://www.marketscreener.com/JOHNSON-JOHNSON-4832/news/Johnson-Johnson-J-J-Offers-4-Billion-Opioid-Litigation-Settlement-29385634/
If completed, the deal would resolve more than 2,000 lawsuits by state and local governments alleging that J&J’s marketing of pain drugs including Duragesic and Nucynta fueled the opioid crisis. J&J sold the U.S. rights to Nucynta to another company in 2015. The suits also allege that J&J is culpable for opioid abuse because of former divisions, which the company sold in 2016, that supplied active ingredients for other opioid manufacturers.
Bloomberg News reported the amount of J&J’s settlement offer Tuesday. The Wall Street Journal reported Tuesday that three major drug distributors were in talks to pay $18 billion to settle the opioid litigation, and that J&J was involved in the discussions to contribute additional money.
Teva Pharmaceutical Industries Inc. also is in talks to settle the opioid lawsuits against it by providing free drugs, according to people familiar with the matter.
A $4 billion settlement would probably please J&J investors, because analysts had expected the company would have to pay more. Wells Fargo had estimated that the opioid litigation could cost the company $5 billion to $10 billion to settle. Wall Street’s concerns about the litigation’s cost have shadowed J&J stock.
In August, an Oklahoma judge ordered J&J to pay $572 million for its role in the state’s opioid crisis, following a trial that began in May. On Tuesday, the judge said he made a miscalculation that overstated the award by more than $100 million, which he said will be reflected in his final order.
J&J said in August it would appeal the judgment and that the judge’s conclusions disregard the company’s compliance with federal and state laws.
The New Brunswick, N.J., company also recently agreed to a $20.4 million settlement of opioid lawsuits brought by two Ohio counties, avoiding a trial in the case scheduled to start next week.
J&J Chief Financial Officer Joseph Wolk told analysts on a conference call Tuesday regarding quarterly financial results that the company was open to settlements of opioid lawsuits like the ones in Ohio.
https://www.marketscreener.com/JOHNSON-JOHNSON-4832/news/Johnson-Johnson-J-J-Offers-4-Billion-Opioid-Litigation-Settlement-29385634/
Subscribe to:
Posts (Atom)