Target $35
Search This Blog
Friday, June 18, 2021
Key senator calls for Woodcock's removal in FDA fallout from Biogen Alzheimer's approval
The FDA’s fallout from its controversial approval of Biogen’s Alzheimer’s drug Aduhelm is rolling on with one pivotal Democratic lawmaker calling on the administration to dethrone the agency’s top decision-maker.
Sen. Joe Manchin (D-W.Va.) called on President Joe Biden in a letter dated Thursday to oust the FDA’s interim commissioner Dr. Janet Woodcock and quickly nominate an “acceptable” permanent chief. The moderate Democrat took aim at the FDA’s decision last week to approve Biogen’s Aduhelm, also known as aducanumab, for Alzheimer’s patients despite intense controversy over whether the treatment actually helps with cognitive decline.
While Biden has yet to name a permanent agency head—a delay that has garnered criticism amid the pandemic—agency veteran Woodcock has been widely considered a top contender. Any future FDA commissioner would need the thumbs up from the Senate, which Democrats narrowly control.
“This approval underscores the fact that more of the same leadership at the agency is not the answer,” Manchin wrote in the letter.
To many, Aduhelm’s wide-ranging FDA label came as a surprise, with some analysts calling it “almost shockingly broad.” The FDA didn’t limit Aduhelm to the early-onset patients studied in clinical trials, but instead gave the medicine the go-ahead for all Alzheimer’s patients.
The FDA's move also went against a resounding no vote from the agency’s own independent advisors late last year. At the very least, the FDA should provide an explanation as to why it went against the advisory committee, which it has yet done, Manchin argued.
Since the approval, three members of the agency’s independent committee have resigned. One of those resignees, Harvard’s Aaron Kesselheim, candidly said in his resignation letter that the Aduhelm nod is “probably the worst drug approval decision in recent U.S. history.”
“The decision to approve drugs without the extra level of scrutiny about their safety and impact on public health has had lasting public health consequences,” Manchin wrote, noting the same rigorous review process for COVID-19 vaccines.
A spokesperson for the FDA and the White House weren’t immediately available for comment.
To be sure, the Aduhelm approval isn’t the only issue the West Virginia senator has taken up with Woodcock. Manchin, along with six other Democrat and Independent senators, called on Biden earlier this year to institute new FDA leadership to address the ongoing opioid crisis. In Manchin’s Thursday letter, the senator said Woodcock has “shown a dereliction of duty by not working to end” the opioid epidemic.
Meanwhile, Manchin isn’t the only one calling for Woodcock, as well as other top FDA officials, to hit the exit.
Earlier this week, consumer advocacy group Public Citizen said Woodcock, as well as CDER Director Patrizia Cavazzoni and Billy Dunn, director of the FDA’s neuroscience unit, should resign following the “indefensible” Aduhelm approval, according to a letter dated Wednesday and sent to the Department of Health and Human Services.
EU loses bid for speedier AstraZeneca vaccine deliveries
Judge orders AstraZeneca to deliver doses by strict deadlines
* EU had asked for more doses to be shipped earlier
* AstraZeneca may have breached contract in reserving doses for UK
* Second legal case will establish whether contract was breached (Adds detail on timing of deliveries)
The European Union on Friday lost its bid to speed up deliveries of AstraZeneca's COVID-19 vaccines, the first of its legal challenges against the drugmaker that rocked the bloc as it scrambled to shore up supplies.
AstraZeneca said the EU had lost its legal case, but European Commission President Ursula von der Leyen said the court ruling supported its view that the Anglo-Swedish pharmaceutical giant had failed to honour its commitments.
The row plunged the EU into crisis earlier this year as states, under pressure to speed up vaccinations, scrambled for shots. Brussels has since largely cut ties with AstraZeneca, choosing not to buy any more of its vaccines for now.
The drugmaker had committed to do its best to deliver 300 million doses to the 27-nation bloc by the end of June, but production delays led it to revise this to 100 million vaccines.
This delayed the EU's vaccination drive as the bloc had initially bet on AstraZeneca to deliver the largest volume, sparking a bitter row and EU legal action to get at least 120 million doses by the end of June.
However, the judge ruled that AstraZeneca must deliver only 80.2 million doses by a deadline of Sept. 27. The drugmaker said it would "substantially exceed" that by the end of June.
The court said in a statement that AstraZeneca must deliver 15 million doses by July 26, another 20 million by Aug. 23 and a further 15 million by Sept. 27, to reach a total of 50 million doses, which are in addition to 30 million that had been given to the EU when the legal case began.
Should it miss the deadlines in the ruling, AstraZeneca would face a penalty of "10 euros ($11.8) per dose not delivered", the judge said, less than the 10 euros per dose per day fine the EU had sought in bringing its legal action.
AstraZeneca will remain bound to do its best to deliver 300 million doses to the EU, and a new hearing is to be held in September when compliance with the contract will be assessed again, the ruling said.
The EU had asked all 300 million doses to be delivered by the end of September, but the court did not set a deadline.
EU data shows the company has already shipped nearly 70 million doses, more than half of which were delivered after the start of the legal proceedings.
This brings AstraZeneca close to already meeting the court's requirement of 80 million doses in total by Sept. 27.
SERIOUS BREACH
An EU lawyer also said the judgment meant that as a proof of best effort, AstraZeneca will have to deliver COVID-19 vaccines from a factory in Britain, if needed to meet its EU commitments.
The company had said it could not immediately deliver to the EU doses from an Oxford BioMedica factory because it had to supply Britain first.
The ruling said that AstraZeneca may have committed a serious breach of the contract by reserving Oxford BioMedica's output for the British market. However a final decision on this will be made in a second legal case.
AstraZeneca said the court had found that the EU had no exclusivity or right of priority over other parties.
"The judgment also acknowledged that the difficulties experienced by AstraZeneca in this unprecedented situation had a substantial impact on the delay," it said in a statement.
"AstraZeneca now looks forward to renewed collaboration with the European Commission to help combat the pandemic in Europe."
The EU last month launched a second legal action against AstraZeneca over an alleged breach of the supply contract, which will continue after the summer. Friday's ruling was over whether AstraZeneca must speed up vaccine deliveries.
Innovation Pharma: New Antiviral Research to be Presented at Military Health System Research Meet
Presentation to be part of the “Development of New Front Line Therapies to Prevent and Treat Non-SARS-CoV-2 Endemic Viral Diseases” Breakout Session
WAKEFIELD, Mass., June 18, 2021 (GLOBE NEWSWIRE) -- Innovation Pharmaceuticals (OTCQB:IPIX) (“the Company”), a clinical stage biopharmaceutical company, today announced that antiviral research related to Brilacidin has been accepted for an Oral Presentation at the 2021 Military Health System Research Symposium (MHSRS), taking place August 23-26, 2021, at the Gaylord Palms Resort and Convention Center, Kissimmee, FL.
The MHSRS is an annual four-day scientific symposium sponsored by the U.S. Department of Defense. The presentation on Brilacidin will be part of the “Development of New Front Line Therapies to Prevent and Treat Non-SARS-CoV-2 Endemic Viral Diseases” Breakout Session. Out of nearly 2,300 submissions, approximately 20 percent were accepted for presentation.
New data supporting Brilacidin’s broad spectrum antiviral properties will be presented at the 2021 MHSRS conference, as well as during a separate oral presentation on Brilacidin to be delivered at the American Society of Virology’s 40th Annual Meeting, to be held in July.
About Brilacidin and COVID-19
Brilacidin is the only non-peptidic defensin-mimetic drug candidate currently in a clinical trial as a treatment for SARS-CoV-2, the coronavirus responsible for COVID-19. Additionally, Brilacidin has shown potent and consistent inhibition in vitro against coronaviruses, alphaviruses and bunyaviruses (with laboratory testing against other viruses also underway), supporting Brilacidin’s potential to be developed as a broad spectrum antiviral. The annual global antiviral drug market is estimated to reach $44 billion by 2026.
A peer-reviewed article in Viruses supporting Brilacidin’s COVID-19 treatment potential can be accessed at the link below.
Bakovic, A.; Risner, K.; Bhalla, N. (et al). Brilacidin Demonstrates Inhibition of SARS-CoV-2 in Cell Culture. Viruses 2021, 13, 271; https://doi.org/10.3390/v13020271
https://www.mdpi.com/1999-4915/13/2/271/
82% of SMA kids treated pre-symptomatically with Zolgensma hit developmental milestones
Swiss Pharma giant Novartis (NOVN: VX) has announced positive new data about Zolgensma (onasemnogene abeparvovec), an essential, one-time treatment and the only gene therapy for spinal muscular atrophy (SMA), from the completed two-copy cohort of the Phase III SPR1NT clinical trial that demonstrates age-appropriate development when used pre-symptomatically and rapid, clinically meaningful efficacy in symptomatic children, even those with severe SMA at baseline.
Highlights of the trial results
The data will be presented at the European Academy for Neurology (EAN) Virtual Congress 2021 (June 19–22), but the main findings are as follows:
- 100% of children (14) treated pre-symptomatically in the SPR1NT two-copy cohort survived without respiratory or nutritional support, and sat independently for ≥30 seconds, including 79% (11/14) within the WHO window of expected normal development;
- 82% treated in STR1VE-EU achieved developmental motor milestones not observed in the natural history of SMA Type 1, including patients with more severe disease;
- 79% (11/14) could stand independently, seven of whom achieved this milestone within the WHO window of normal development;
- 100% of patients (14/14) met the secondary endpoint of survival without ventilatory support of any kind at 14 months of age, versus only 26% of patients in the Pediatric Neuromuscular Clinical Research (PNCR) natural history cohort; and
- More than 1,200 patients have now been treated with Zolgensma globally across clinical trials, managed access programs, and in the commercial setting.
SPR1NT trial results ‘nothing short of extraordinary’
“With more than 1,200 children now treated, these data presented at EAN further reinforce the life-changing benefit of a one-time treatment of Zolgensma,” said Dr Shephard Mpofu, senior vice president, chief medical officer, Novartis Gene Therapies.
“When treated with Zolgensma prior to the onset of symptoms, not only did all patients survive, but were thriving - breathing and eating on their own and sitting independently, with many standing and walking. When you consider these newborns would go on to develop severe symptoms of SMA Type 1, a devastating, progressive disease that robs children of the ability to talk, eat, sit up and even breathe, findings from the SPR1NT trial are nothing short of extraordinary,” Dr Mpofu opined.
‘Most expensive drug in the world’
Zolgensma, which gained conditional approval in Europe in May 2020 and in the USA a year earlier, has a reported list price of £1.8 million ($2.5 million) per dose, and is labelled the most expensive drug in the world. The other treatment available to treat children with SMA is Biogen’s (Nasdaq: BIIB) much older drug Spinraza (nusinersen), but Novartis’ drug is thought to be more cost-effective than Spinraza over the long term. Also available is Roche’s (ROG: SIX) Evrysdi (risdiplam). Zolgensma is forecast to generate global sales of $2.5 billion by 2025 and lead the spinal muscular atrophy (SMA) market, according to data and analytics company GlobalData.
'Not Going Away' - Retail Trading Continued To Surge During First Half Of 2021
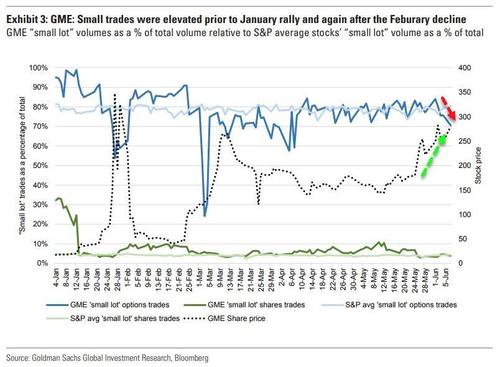
Last week, we shared some new research from Goldman Sachs that confirmed what many had observed over the past year: the flood of newly-minted retail traders seen over the past year and a half has transformed retail trading activity from a contrarian to a leading indicator.
In fact, the newfound dominance of retail traders has taken an old market dynamic and flipped it on its head: in contemporary markets, it's retail traders who generate buzz in a stock by driving it higher, drawing the attention of institutional traders, before the "dumb" money rotates out, leaving the "smart" money holding the bag. The dynamic has been perhaps the most pronounced with GME.
Institutional traders have taken notice of this newfound retail "edge", which is why firms like Cindicator Capital are looking to hire a longtime "WallStreetBets" vet instead of an institutional trader. The firm's job posting made the media rounds because of its ask that applicants have "at least 1,000 of Reddit's goodwill karma points." The firm is offering a salary of $200K plus bonuses (along with a place in a Burning Man camp).
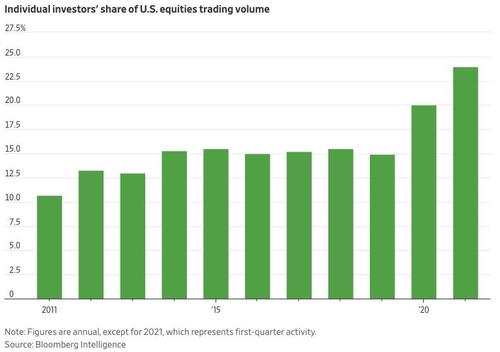
And while that posting likely elicited plenty of scoffs from the industry, a WSJ report published Friday suggests that the firm might be on to something, since the explosion of retail trading activity that emerged last year has only intensified during the first half of 2021. Citing an estimate from JMP Securities, WSJ said new brokerage accounts opened by individual investors have already roughly matched the total opened throughout 2020.
What's more, individual investors have poured a net $140.57 billion into US stocks this year, according to data from Vanda Research’s VandaTrack. That's up roughly 33% from the same period a year ago.
Thanks to zero-commission trading and the advent of payment for order flow, has sent retail's share of average daily US equity-market turnover to 20%, double its level from a decade ago.
Larry Tabb, an analyst who focuses on market structure, warned that he expected individual investors' elevated levels of trading activity to continue for the foreseeable future.
"They definitely have influence, and they’re not going away," he said. "We think for the next year or so, 18% to 22% of the market will be driven by retail."
Data shows that retail traders are mostly focused on the short term, and they tend to move into and out of stocks with great force.
Take, for example, a basket of meme stocks that feature prominently on Reddit’s WallStreetBets platform. Data from VandaTrack shows that investors moved into the basket—which features companies including GameStop, AMC and BlackBerry Ltd. —quickly in January, before largely abandoning the trade only three weeks later.
Inflows into the basket have surged again in recent weeks, but already, data shows, the tide is turning. The price of the basket has fallen 17% over the past week, Vanda Research said in a note Wednesday morning, as flows into the stocks have also begun to slow.
"This is changing the way that one potentially trades these spaces—gone are the days when you can buy and hold a small-cap name and hope it yields 50% over time. It almost does that now in a matter of days," said Viraj Patel, global macro strategist for Vanda Research. "Market timing is the best friend of the app trader in this space."
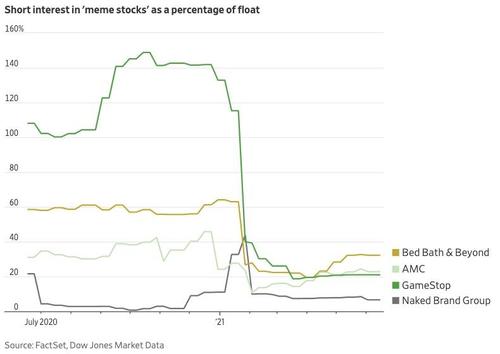
This has made short-sellers extremely wary, causing short interest in these stocks to slump.
The surge in retail trading has been a boon for electronic market-makers like Citadel and Virtu. It's also been a boon for banks and other operators of "dark pools", as these firms increasingly favor private trading venues to executing trades on the New York Stock Exchange. In January, the amount of trading volume in the US market started happening off exchanges climbed to a record 47%, data from Rosenblatt Securities shows. Certain stocks have seen off-exchange trading volume soar to nearly 80% of trading for Sundial Growers. In May, the figure for AMC was 57%.
"There were many days in December and January in which off-exchange trading was greater than 50% of total volume," said Justin Schack, partner at Rosenblatt.
Why is this privacy so important to the market makers who have been the ultimate winners from the retail trading explosion? While regulators have acquiesced to the practice thanks to hollow arguments about price improvement, studies have shown that this appears to be a case of penny-wise, pound-foolishness. In fact, as Themis Trading explained in recent commentary to the House Financial Services Committee, PFOF increases overall costs for all investors.
Oncology biotech Cyteir Therapeutics sets terms for $126 million IPO
Cyteir Therapeutics, an early stage oncology biotech developing small molecule therapies that inhibit DNA damage repair, announced terms for its IPO on Monday.
The Lexington, MA-based company plans to raise $126 million by offering 7.4 million shares at a price range of $16 to $18. At the midpoint of the proposed range, Cyteir Therapeutics would command a fully diluted market value of $618 million.
Cyteir Therapeutics' lead candidate, CYT-0851, is a novel, oral small molecule inhibitor of homologous recombination, a DNA repair pathway critical for the survival of some cancers. CYT-0851 is currently in a Phase 1/2 trial in adult patients with hematologic malignancies and solid tumors. The company states that it has observed encouraging preliminary single agent activity and may initiate a registrational trial of the candidate in 2022, if warranted by the Phase 1/2 data and subject to FDA agreement. Cyteir Therapeutics also plans to initiate dosing in a Phase 1/2 trial of CYT-0851 in combination with various standard-of-care therapies in the 2H21.
Cyteir Therapeutics was founded in 2012 and plans to list on the Nasdaq under the symbol CYT. J.P. Morgan, Morgan Stanley, BofA Securities, and Wedbush PacGrow are the joint bookrunners on the deal. It is expected to price during the week of June 14, 2021.