Data presented at ASCO 2019 showed 57% of patients
alive at three years vs. 43.5% on placebo
AstraZeneca has presented three-year overall survival (OS) results from the Phase III PACIFIC trial of Imfinzi (durvalumab) in unresectable, Stage III non-small cell lung cancer (NSCLC) during the 2019 American Society of Clinical Oncology (ASCO) Annual Meeting in Chicago.
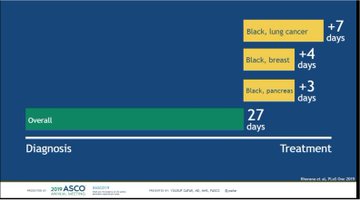
These latest results show a durable and sustained OS benefit in patients with unresectable, Stage III NSCLC who had not progressed following concurrent chemoradiation therapy (CRT), a previous standard-of-care (SoC) treatment. The OS rate was 57% at three years for patients receiving Imfinzi vs. 43.5% for placebo following concurrent CRT. Median OS was not yet reached with the Imfinzi arm vs. 29.1 months for placebo.
Dave Fredrickson, Executive Vice President, Oncology Business Unit said: “These findings for Imfinzi are another example of our focus on bringing long-term survival benefits to patients who still have a chance of being cured. These three-year survival results further establish the PACIFIC regimen as the standard of care for these patients, and we are optimistic this survival trend will continue as we move towards the five-year landmark in this curative-intent setting.”
Results build on the primary two-year OS analysis that was published in The New England Journal of Medicine in September 2018 and demonstrated a significant OS benefit for treatment with Imfinzi vs. placebo after CRT, regardless of PD-L1 expression. The primary analysis showed Imfinzi reduced the risk of death by 32% (HR 0.68, [99.73% CI, 0.47-0.997], p=0.0025).
With the additional year of follow up, the latest results for Imfinzi showed consistent and durable efficacy, maintaining a 31% reduction in the risk of death vs. placebo after CRT (HR 0.69, [95% CI 0.55-0.86]).