By Adam Andrzejewski, author of OpenTheBooks substack
California Governor Gavin Newsom has quietly solicited millions of dollars in campaign donations from state vendors, key people, employees, or their affiliated corporate political action committees.
While progressives decry corporate money in politics, Governor Gavin Newsom has embraced the highly unethical practice of soliciting campaign cash from state contractors.
Our auditors at OpenTheBooks.com found 979 state vendors who gave $10,561,828 in political donations to Newsom during his 2010, 2018, recall election, and 2022 election cycles. Meanwhile, these companies reaped $6,201,978,173 in state payments.
These donations represented the equivalent of more than 40-percent of the current cash on hand – $24 million – in the governor’s campaign committee as of 08/01/2022, according to disclosures.
We created an interactive map displaying by ZIP Code all of the governor’s campaign contributions—by name and employer, amount donated, when, and where located – across America. Just click a pin (ZIP Code) and scroll down to see the results that render in the chart beneath the map.
Mapping campaign donations: Newsom For California Governor 2010, 2018, 2022:
We reached out to Newsom and his press secretary Daniel Lopez requesting comment — giving the governor a chance to defend his fundraising practice. We will update the piece if the Newsom responds on the record.
Since 1940, however, individuals and entities negotiating or working under federal contracts have been prohibited from giving political cash to federal candidates, parties, or committees. In California, however, this political patronage is perfectly legal (at least for now).
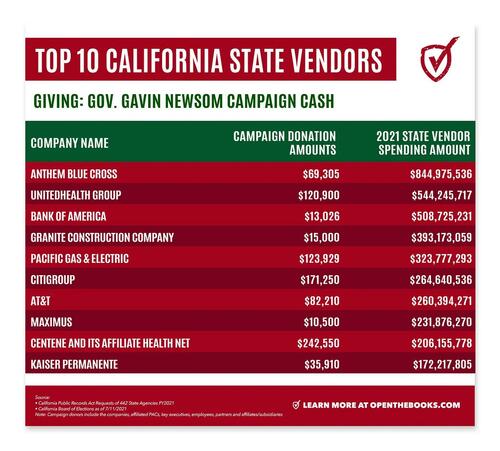
Here are some of the companies who gave campaign cash to Gov. Newsom and separately received state payments. In all these examples, the donations came from the organization itself or its executives, employees, subsidiaries, partners, or political action committees during Newsom’s 2010, 2018 and 2022 election cycles.
I. MAJOR HEALTH CARE COMPANIES – Gave $691,615 in campaign donations and received $1.9 billion in state payments.
Anthem Blue Cross (health insurance provider) received $844,875,535 in state payments while donating $69,305 during Newsom’s 2018 and 2022 elections— $40,000 of that was during the 2022 cycle.
UnitedHealth Group (managed health care and insurance provider) received $544,245,717 in state payments while donating $120,900 between the 2018 and 2022 cycles — $62,000 was donated in the 2022 cycle. Even the Chief Compliance Officer Joy Hia donated $500 to the 2022 campaign.
Centene Corporation (Fortune 500 managed care company) and Health Net, LLC, a major subsidiary, which provides health plans for those with Medicare and Medicaid, received $206,155,778 in state payments while donating $242,550. The company itself donated $121,800 while then-CEO Michael Neidorff gave $120,400 between 2018 and 2022. Not included is an additional $120,400 from wife Noemi Neidorff. Michael Neidorff passed away on April 7, 2022.
Kaiser Permanente received $172,217,805 while employees donated $35,910 including the vice president of government relations, Gary Cohen ($5,000 | 2018 election). Blue Shield of California received $74,283,100 in state payments while donating $102,550 including $70,200 from the company and $32,350 from key executives and employees. Masimo Corporation, a health-tech company, received $3,820,654 in state payments and gave $120,400 to Newsom’s 2018 and 2022 races – half of the donations came in 2022.
II. MAJOR UTILITY COMPANIES – Gave $405,601 in campaign donations and received $430,416,420 in state payments
Pacific Gas & Electric Company (PG&E) based in San Francisco heavily criticized for its role in the California wildfires and recently came out of bankruptcy. The company received $323,777,292 in state payments (FY2021) and gave $123,929 in donations for the 2018 election. These donations included five and six figure gifts from five C-suite execs including CEO Geisha Williams ($10,000). Due to continuing scandals, Newsom stopped taking donations from PG&E after his election in 2018. The company also gave $358,000 between 2011-2018 to Newsom’s wife, Jennifer Siebel Newsom’s charity.
Edison International, with subsidiaries Southern California Edison and consultancy, Edison Energy, LLC, received $100,575,389 in state payments and gave $67,850 in campaign donations through the company, key employees, and staff. Additionally, the company’s trade association, Edison Electric Institute, donated $15,000 to Newsom’s 2018 race.
Calpine Corporation, the largest generator of electricity from natural gas and geothermal sources in the United States with 33 facilities in California received $3,134,154 in state payments and gave $109,822 in campaign donations. CEO Thad Hill gave $10,000 and other top execs gave $12,500 to Newsom.
California Water Service Company (Cal Water) received $2,121,724 in state payments and gave $94,000 between Newsom’s 2018 and 2022 campaigns.
California American Water Company, a subsidiary of American Water – the largest publicly-traded water and wastewater utility in the U.S. – received $807,861 in state payments and gave $10,000 through their employees PAC in the 2018 election. American Water also gave at least $5,000 to the governor’s wife’s charity in 2019.
III. MAJOR TELECOM COMPANIES – Gave $241,959 in campaign donations and received $420.3 million in state payments
AT&T received $260,394,271 in state payments and donated $82,210 to Newsom’s 2018 campaign. Public affairs executive Ken McNeely donated $17,000 between the 2010 and 2018 campaigns and also co-chaired the 2019 Flip The Script Gala for The Representation Project, a non-profit founded by Gavin Newsom’s wife Jennifer Siebel Newsom.
Verizon received $130,184,875 in state payments and donated $46,700 with the majority of the donations ($45,000) through the company itself.
Telrite Holdings (Life Wireless) received $9,102,033 in state payments and donated $10,000 to the 2018 campaign. California operates its LifeLine, the free phone program for low-income residents.
Comcast received $20,627,409 in state payments and donated $103,049. The company itself gave $29,200. Employees gave $73,849 including senior executive vice president David Cohen, based in Philadelphia, who gave $29,200 to the 2018 election. The company also gave at least $15,000 to The Representation Project (Newsom’s wife’s charity).
IV. BIG LAW – top nine firms gave $198,142 in campaign donations and received $28.6 million in state payments
We found nine law firms that reaped $28,615,984 in state payments and gave $198,142 in campaign cash to the governor. These donations came the law firms, principals, partners, key employees, or staff.
Top donors included Nixon Peabody who gave $94,272 and received nearly $2.2 million in payments; Perkins Coie who gave $36,400 and received $1.4 million in payments; and Orrick Herrington & Sutcliffe donated $31,710 to Newsom and received $4.3 million in state payments. Others prominent firms backing the governor and receiving millions of dollars in state payments included: Loeb and Loeb ($11,850) and Munger Tolles & Olson ($5,750).
V. BIG BANKS – Gave $186,836 in campaign donations and received $781 million in state payments.
- Bank of America received $508,725,231 in state payments while donating $13,026. $10,000 came through their state and federal political action committee in 2018 while $3,026 was donated by employees.
- Citigroup received $264,640,535 in state payments while donating $147,050. CEO William Mills donated $24,600 to Newsom’s 2018 race. The Citigroup state political action committee donated $110,600 between the 2018 and 2022 races.
- Wells Fargo received $7,578,648 in state payments while donating $26,760 to Newsom’s 2018 race – staff gave $21,760 and the bank’s PAC donated $5,000.
OTHERS
Two railway companies with quasi-marketplace monopolies reaped nearly $47,505,454 in state payments during fiscal year 2021 and gave $112,400 in campaign donations to Newsom since 2010. BNSF Railway Company received $40,411,142 in state payments and gave $26,200 between the 2018 and 2022 campaigns. Union Pacific Railway Company received $7,094,312 in state payments and gave $86,200 in the 2018 and 2022 elections.
Ten California based Native American tribes donated over $841,800 to Newsom’s campaign fund and their benefits are harder to quantify. Although those tribes received $8,753,578 in state payments (FY2021), their casinos are a highly regulated state business (in addition to many other interests before the state).

Jennifer Siebel Newsom’s non-profit loophole
While the governor was soliciting state vendors for campaign donations, Mrs. Newsom, the first partner, Jennifer Siebel Newsom, solicited state vendors for donations to her charity, The Representation Project.
Major corporations with state contracts or business before the state gave the charity five and six figure gifts. The Sacramento Bee and Washington Post previously identified the companies and today we know just how much those corporations reaped in state agency payments. (23 and Me is the only donor that wasn’t on the state vendor list, however, they had an interest in 2021 state legislation regulating the use of consumer genetic data.)
IRS 990 informational returns for The Representation Project show that Siebel-Newsom took $1.5 million in salary from 2013-2021 and another $1.6 million in payments to her private company, Girls Club Entertainment since 2012.
Summary
In all the examples identified above, no quid-pro-quo is alleged or implied; however, the patterns are troubling. In fact, the individual transactions are legal at arm’s length.
But that’s precisely the problem. Politicians preside over, in essence, a legalized money-recycling scheme aimed at monetizing incumbent political power.
Newsom didn’t answer our question as to whether soliciting state contractors for campaign cash was ethical. Meanwhile, California residents are left with skyrocketing taxes and an increasingly bleak future.
We can blame Governor Newsom, but he is just a reflection of today’s broken culture of public service.
Methodology: We matched companies donating to Newsom For California 2010, 2018, 2022, as disclosed by the California State Board of Elections, to state payment transactions from fiscal year 2021– which we compiled through 442 California Public Record Act requests. To the extent that the information contains government errors, our report will reproduce those errors. No quid pro quo or illegal activity by any elected official, company or individual referenced in this piece is implied or intended. Gavin Newsom was elected governor in 2019.
We have requested comment from the governor and The Representation Project, the first partner’s public charity.
ADDITIONAL READING


