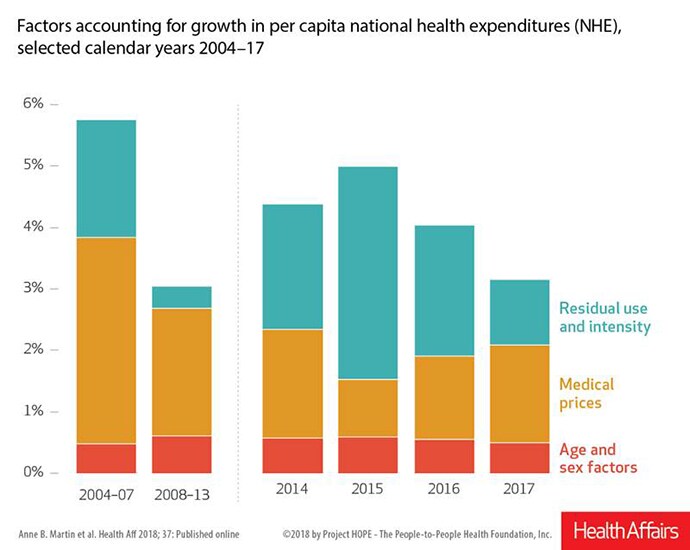
US spending for physician and clinical services rose 4.2% in 2017, the smallest increase seen since 2013, according to a new report. This continues a recent ebb in the growth rate for total national healthcare expenses, which followed a jump in 2014 and 2015 due to expansion of Medicaid and private insurance coverage.
Total US health spending experienced a bump in annual growth rates in 2014 (5.2%) and 2015 (5.8%), compared with annual increases of between 3% and 4% seen from 2011 to 2013, CMS said.
Around 2014, many states expanded their Medicaid programs, while at the same time, public and private insurers wrestled with the cost of new drugs for hepatitis C. By 2016, however, the Medicaid expansion was largely established and many people had been treated for hepatitis C, thus the growth rate for total US health spending that year slowed to a 4.8% increase.
In 2017, national healthcare spending rose by just 3.9% to a total of $3.49 trillion.
Spending on physician and clinical services followed a similar pattern, with annual gains between 2.2% and 4.6% from 2011 to 2014, CMS said. The growth rate then hit 6.0% in 2015, slowed to 5.6% in 2016, and slipped again to 4.2% for last year. Spending for physician and clinical services totaled $694.3 billion last year, CMS said.
On a call with reporters, Aaron Catlin, deputy director in the National Health Statistics Group in CMS’ Office of the Actuary, said there are a number of factors that could play into the drop in growth rate for physician and clinical services for which the agency hasn’t created specific estimates.
In response to a question from a reporter, Catlin said high-deductible health plans might play a role in the decrease in intensity of services. (These plans make their customers directly pay a larger share of some medical bills, giving them an incentive to reconsider some visits or treatments.) He also noted that there has been a shift in site of care, with some physician services shifted to freestanding and other clinical settings.
Drug Spending
The recent spike and decline in spending growth has been even more pronounced for prescription drugs. Annual increases in this spending ranged from 0.2% to 2.3% in 2011 to 2013. A 12.4% spike occurred in 2014, due in part to the introduction of costly sofosbuvir (Sovaldi), a hepatitis C antiviral drug sold by Gilead Sciences Inc.
The rate of growth has slowed since, dropping to 0.4% last year, with retail prescription drug spending totaling $333.4 billion. The slower growth rate reflects a continued shift to lower-cost generic drugs, slower growth in the volume of some high-cost drugs, declines in generic drug prices, and lower price increases for existing brand-name drugs, CMS said.
These cost-saving steps for pharmaceutical purchases were, however, balanced by the continued introduction of new medicines, which often command high prices. In the new report, CMS noted that there were 46 new drug approvals last year, more than double the 22 approvals in 2016.
The 2017 introductions included 13 new drugs approved for cancer, CMS said. Of the new drugs introduced in 2017, 15 were “first in class,” including drugs for eczema and multiple sclerosis. Eighteen new drugs were intended for rare diseases and are often referred to as “orphan drugs,” CMS said.
CMS officials also reported that:
- Hospital spending rose 4.6% to $1.1 trillion, less than the 5.6% growth seen in 2016.
- Private health insurance spending increased 4.2% to $1.2 trillion in 2017, less than the 6.2% growth seen in 2016. CMS said this deceleration reflects both slower growth in medical benefits and a decline in taxes resulting from the suspended collection of a health insurance provider fee in 2017.
- Medicare spending grew 4.2% to $705.9 billion in 2017, little changed from 2016 when spending grew 4.3%. Growth in fee-for-service Medicare spending was 1.4% in 2017 compared to 2.6% in 2016, while spending for Medicare private health plans rose 10.0% in 2017 compared to 8.1% in 2016.
- Medicaid spending growth slowed in 2017, increasing 2.9% to $581.9 billion following growth of 4.2% in 2016.
- Out-of-pocket spending by consumers, including copayments, deductibles, and spending not covered by insurance, grew 2.6% to $365.5 billion in 2017, which was slower than the 4.4% growth in 2016.
The CMS officials ended their report by highlighting the overall trend toward having healthcare costs consume more of the nation’s economy, although there was a slight drop last year in its share. Healthcare spending accounted for 17.9% of GDP last year, as opposed to 18.0% in 2016.
Last year was the first since 2013 that healthcare’s share of the gross domestic product did not increase. “2014 and 2015 were marked by insurance coverage expansion and expensive new drug treatments that produced faster growth in health care spending, while 2016 was affected by low economic growth,” the CMS officials said.
The CMS officials noted that health care spending growth averaged 4.3% per year between 2008 and 2017, compared with an average annual rate of 7.3% in 1998 through 2007.
Even so, healthcare spending’s share of GDP increased by 1.6 percentage points from 2008 to 2017, compared with a 2.6-percentage-point increase from 1998 to 2007, CMS said.
“For a health sector that now accounts for nearly one-fifth of the US economy, future increases in health care expenditures will likely lead to policy decisions focused on affordability and sustainability,” the CMS officials wrote.
The authors have disclosed no relevant financial relationships. The opinions expressed in the analysis are the authors’ and not necessarily those of CMS.
Health Aff. Published online December 6, 2018.
Abstract
CMS. Published online December 6, 2018.
Report