The more we learn about the novel coronavirus (COVID-19), the more
unknowns seem to arise. These ever-emerging mysteries highlight the
desperate need for more data to help researchers and physicians better
understand—and treat—the extremely contagious and deadly disease.
Researchers at Northwestern University and Shirley Ryan AbilityLab in Chicago have developed a novel
wearable device
and are creating a set of data algorithms specifically tailored to
catch early signs and symptoms associated with COVID-19 and to monitor
patients as the illness progresses.
Capable of being worn 24/7, the
device
produces continuous streams of data and uses artificial intelligence to
uncover subtle, but potentially life-saving, insights. Filling a vital
data gap, it continuously measures and interprets coughing and
respiratory activity in ways that are impossible with traditional
monitoring systems.
Developed in an engineering laboratory at Northwestern and using
custom algorithms being created by Shirley Ryan AbilityLab scientists,
the devices are currently being used at Shirley Ryan AbilityLab by
COVID-19 patients and the healthcare workers who treat them. About 25
affected individuals began using the devices two weeks ago. They are
being monitored both in the clinic and at home, totaling more than 1,500
cumulative hours and generating more than one terabyte of data.
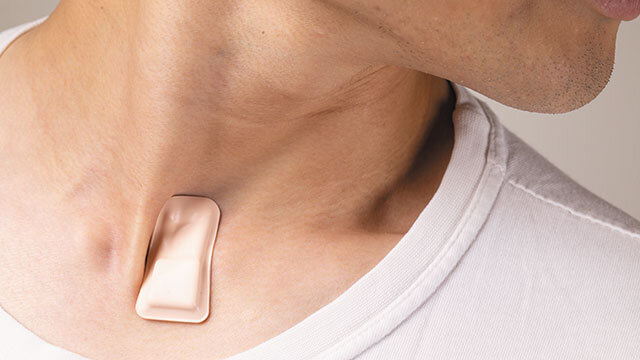
About the size of a postage stamp, the soft, flexible, wireless, thin
device sits just below the suprasternal notch—the visible dip at the
base of the throat. From this location, the device monitors coughing
intensity and patterns, chest wall movements (which indicate labored or
irregular breathing), respiratory sounds, heart rate and body
temperature, including fever. From there, it wirelessly transmits data
to a HIPAA-protected cloud, where automated algorithms produce graphical
summaries tailored to facilitate rapid, remote monitoring.
“The most recent studies published in the
Journal of the American Medical Association
suggest that the earliest signs of a COVID-19 infection are fever,
coughing and difficulty in breathing. Our device sits at the perfect
location on the body—the suprasternal notch—to measure respiratory rate,
sounds and activity because that’s where airflow occurs near the
surface of the skin,” said Northwestern’s John A. Rogers, who led the
technology development.
“We developed customized devices, data algorithms, user interfaces and
cloud-based data systems in direct response to specific needs brought to
us by frontline healthcare workers. We’re fully engaged in contributing
our expertise in bioelectronic engineering to help address the
pandemic, using technologies that we are able to deploy now, for
immediate use on actual patients and other affected individuals. The
measurement capabilities are unique to this device platform—they cannot
be accomplished using traditional watch or ring-style wearables that
mount on the wrist or the finger.”
“We anticipate that the advanced algorithms we are developing will
extract COVID-like signs and symptoms from the raw data insights and
symptoms even before individuals may perceive them,” said Arun
Jayaraman, a research scientist at Shirley Ryan AbilityLab, who is
leading the algorithm development. “These sensors have the potential to
unlock information that will protect frontline medical workers and
patients alike—informing interventions in a timely manner to reduce the
risk of transmission and increase the likelihood of better outcomes.”
Continuous monitoring from hospital to home
The mysterious ways that COVID-19 affects the body seem to get
stranger and stranger. Many patients’ symptoms fully disappear before
they suddenly and unexpectedly begin deteriorating—sometimes within a
matter of hours. Other patients have recovered and tested “negative” but
later test “positive” again.
The unknowns underscore the need for continuous patient monitoring to
ensure that physicians can intervene at the slightest sign of trouble.
The Northwestern and Shirley Ryan AbilityLab teams’ device provides
around-the-clock monitoring for COVID-19 patients and those exposed to
them.
“Having the ability to monitor ourselves and our patients—and being
alerted to changing conditions in real time—will give clinicians a new
and important tool in the fight against COVID-19,” said Dr. Mark Huang, a
physician at Shirley Ryan AbilityLab, who has worn the sensor. “The
sensor also will offer clinicians and patients peace of mind as it
monitors COVID-like symptoms, potentially prompting earlier intervention
and treatment.”
The device can monitor hospitalized patients and then be taken home
to continue 24/7 supervision. The real-time data streaming from patients
gives insights into their health and outcomes that is currently not
being captured or analyzed by traditional monitoring systems.
“Nobody has ever collected this type of data before,” Rogers said.
“Earlier detection is always better and our devices provide important
and unique capabilities in that context. For patients who have
contracted the disease, the value is even more clear, as the data
represent quantitative information on respiratory behavior, as a
mechanism to track the progression and/or the effects of treatments.”
“This opens up new telemedicine strategies as we won’t have to bring
in patients for monitoring,” Jayaraman said. “Physicians can potentially
review the patients’ data for hours, days or weeks, immediately through
a customized graphical user interface to a cloud data management system
that is being set up for this purpose, to see an overall image of how
the patient is doing.”
Although the wearable device is currently unable to measure blood
oxygenation levels, which is an important component of lung health, the
team plans to incorporate this capability in its next round of devices.
The Rogers lab has already suc … ntensive care units. Rogers believes they can easily apply that research to the COVID-tailored devices.
Warning system for the most at-risk
Not only can the device monitor the progress of COVID-19 patients, it
could also provide early warning signals to the frontline workers who
are most at risk for catching this remarkably infectious disease. The
device offers the potential to identify symptoms and to pick up trends
before the workers notice them, thereby providing an opportunity to
engage in appropriate precautionary measures and to seek further testing
as quickly as possible.
“People with obvious, severe symptoms are going to the hospital,
being tested or being told to self-isolate,” Jayaraman said. “For those
who have symptoms they perceive as mild or seasonal allergies, there is
no warning system. They could be in contact with others and unknowingly
spread infection.”
Assessing efficacy of new therapeutics
As researchers rush for a COVID-19 cure, physicians have been trying
exploratory, sometimes unproven, treatments to help their patients. This
is another area where Rogers’ and Jayaraman’s device can play a role.
“Early reports of certain proposed treatments suggest that they can
eliminate coughing symptoms more quickly than a placebo,” Rogers said.
“Nobody, however, is quantifying certain key symptoms, such as
coughing—duration, frequency, amplitude, sounds, etc. Our device allows
for precision measurement of this essential, yet currently unquantified,
aspect of the disease.”
In the future, this sensor package could help researchers and physicians quantify which therapeutics are working best.
“At the simplest level, our systems allow assessments based on data,
in a quantitative way, without relying on human judgment of whether a
patient is coughing more or less,” Rogers said.
Device initially conceived for stroke patients
The new device builds on recent research from a collaboration between
Rogers’ and Jayaraman’s labs, first published on the cover of the
February 2020 issue of
Nature Biomedical Engineering, with a
focus on monitoring swallowing and speech disorders in patients
recovering from stroke. These sensors work by precisely measuring
vibratory signatures from the throat and chest. By measuring vibrations
rather than acoustics, the team avoids noise from background sounds and
it bypasses privacy issues.
In response to requests and inquiries from the medical community,
Rogers and Jayaraman realized they could use this technology to measure
the vibratory signatures of COVID-like symptoms, including chest wall
movements and cough.
Jayaraman’s team is developing custom signal processing and
machine-learning algorithms to train the device how to recognize coughs
in the data.
“As the algorithm becomes smarter, our hope is that it will begin to
discriminate among which coughs are COVID-like and which are from
something more benign,” Jayaraman said. “The most basic approach,
already deployed on COVID-19 patients and health care workers, simply
counts coughs and their intensity.”
More advanced analytics packages will be available within the next few weeks.
Bypassing already-stressed supply chains
Thanks to a generous gift from Northwestern University trustees
Kimberly K. Querrey and Louis A. Simpson, Rogers and his team are able
to respond quickly to requests for devices. Leveraging a set of
manufacturing tools available in the
new Simpson Querrey Biomedical Research Building in Chicago, the team
is already producing dozens of devices per week. Rogers estimates that
his team could produce up to hundreds of devices per week—all in house,
largely bypassing the need for external vendors and complex supply
chains.
“Quickly developing new technologies internally has never been more
crucial,” Querrey said. “This work proves the power of STEM and why it’s
so critical to the University and beyond to have world-class
researchers like John. I am so proud of John and his team, while working
remotely, for thinking outside the box and using their collaborations
to help protect our healthcare workers. We are excited to be able to
develop these devices within the University and get them in the hands of
those needing them most. The ability to measure vibratory signatures
could really help with early detection of COVID-19.”
“This crucial philanthropic support has allowed us to develop and
deploy the devices and an associated software infrastructure almost
immediately, within days, after we started receiving requests from the
medical community—without waiting for external vendors, most of which
are mostly shut down with the stay-at-home orders,” Rogers said. “In
this way, we avoid already-stressed supply chains. We just do it
ourselves.”
Comfortable and easy to use
In mid-March, Kelly McKenzie felt foggy and developed a low-grade
headache. Having recently returned from a work-related trip overseas,
she assumed it was jetlag. But as her symptoms progressed to include
cough and congestion, she started to worry. Although her symptoms were
not severe enough to seek COVID-19 testing, she knew she should
self-isolate.
“Between my international travel and the symptoms, my director and I
decided it was best for me to stay home from work, so I wasn’t bringing
anything contagious into the hospital,” said McKenzie, who is a research
physical therapist at Shirley Ryan AbilityLab.
McKenzie joined the pilot study to test the device and train the
algorithm with her symptoms. After wearing the sensor around the clock
for a week, she was amazed by the comfort of the soft silicone material
and ease of use. Wearers simply charge the device, put it on and it
immediately begins to work—streaming real-time data to a smartphone or
tablet.
“When you first put it on, you can feel it just because it’s new and
different,” McKenzie said. “But after you have worn it for a while, you
don’t even notice it.”
Because it is fully encased without wires, electrodes, charge ports
or removable batteries, the device can be worn while exercising or in
the shower. It turns out this also is important for sterilization and
reuse.
“This is absolutely critical for use in the context of this extremely
contagious disease,” Rogers said. “Because it is fully sealed in a soft
biocompatible silicone material, it can be completely immersed in
alcohol, and then exposed to a gas-based system for rigorous
sterilization. If there were exposed regions, or plugs or ports or other
physical interfaces, the device would not be relevant for this
application.”
What’s next?
In the coming weeks, the Northwestern and Shirley Ryan AbilityLab
teams will continue collecting patient data to strengthen their
algorithms—through deployments both in the clinic and at home. They also
are responding to other requests for access to the technology, across
the medical complex in Chicago. Additional deployments are starting now.
Rogers and Jayaraman also are examining data from patients recovering
from COVID-19, attempting to determine when they are no longer
contagious. Some of the patients wearing the device have been dismissed
from the acute-care hospital and are rehabilitating at Shirley Ryan
AbilityLab. In the future, this device could help determine whether
post-COVID patients still have minor, perhaps imperceptible symptoms.
Rogers hopes the device will not just tell physicians how to best
treat COVID-19 but also inform researchers about the nature of the virus
itself.
“The growing amount of information and understanding around COVID-19
as a disease will be critically important to containing and treating the
current outbreak as well as those that might occur in the future,” he
said. “We hope, and we believe, that these devices may help in these
efforts by identifying and quantifying characteristics and essential
features of cough and respiratory activity associated with this
disease.”
To accelerate the deployment of this device, the team recently
launched a lean engineering-centric company, Sonica Health, based on
intellectual property jointly developed by Northwestern and the Shirley
Ryan AbilityLab and licensed through Northwestern’s Innovation and New
Ventures Office. Exploring use of the device for the COVID-19 response
is supported by the Biomedical Advanced Research and Development
Authority (BARDA), part of the Office of the Assistant Secretary for
Preparedness and Response at the U.S. Department of Health and Human
Services.
BARDA invests in the innovation, advanced research and development,
acquisition and manufacturing of medical countermeasures—vaccines,
drugs, therapeutics, diagnostic tools and non-pharmaceutical products
needed to combat health security threats. To date, 54 BARDA-supported
products have achieved regulatory approval, licensure or clearance.
DRIVe (Division of Research, Innovation and Ventures) within BARDA,
catalyzes the development of innovative products and approaches, like
the Sonica Health technology, with the aim of solving major health
security challenges.
