Whole-slide imaging is now a validated component of modern pathology practice, supported by increasingly robust foundation models that generalize across stains and scanners and by updated Clinical Laboratory Improvement Amendments (CLIA) guidance that clarifies digital and remote workflows (see part 1). Just as importantly, the same biopsy can now support triage, prognosis, molecular testing, and even treatment selection without additional tissue handling.
This convergence of validated digital slides, stronger AI backbones, and clearer regulatory footing sets the stage for the next shift: AI that doesn’t just detect cancer but informs treatment decisions.
Here is what’s cleared or designated today and how it fits into oncology practice.
First Image-Based Treatment Biomarker
In August 2025, the FDA granted de novo authorization to ArteraAI Prostate. ArteraAI is the first AI-powered digital pathology tool cleared to provide both prognostic and predictive information in localized prostate cancer, creating a new product-code category for similar tools.
It analyzes biopsy whole-slide image plus clinical variables to estimate 10-year metastasis risk, prostate cancer-specific mortality, and the likelihood of benefit from treatment intensification (such as adding androgen deprivation therapy or androgen receptor pathway agents to radiation). It is included in NCCN Prostate (category 2A), and Medicare payment is established (effective January 1, 2024). Notably, FDA’s order includes a predetermined change control plan, allowing Artera to add scanner compatibility without full resubmission.
Consider a patient with high-risk localized prostate cancer preparing to start radiation. Traditional inputs include Gleason grade, prostate-specific antigen, clinical stage, and in some cases genomic classifiers. ArteraAI adds a morphology-derived estimate of treatment benefit, supported by validation anchored in datasets linked to trials such as STAMPEDE.
The shift is subtle but important. Instead of asking, “Is this patient high risk?” you can begin asking, “Is this tumor biologically likely to benefit from intensification?” This approach does not replace genomics or clinical judgment. It augments them with slide-derived biology.
The UK Vanguard Path program will evaluate how well the predictions made by ArteraAI align with real-world outcomes among more than 4000 men treated for prostate cancer over at least 5 years, measuring the AI’s real-world impact on treatment decisions and timelines.
Detection-Assist Tools
Before predictive AI tools such as ArteraAI come assistive AI tools such as Paige and Ibex, which function as safety nets within routine pathology workflows.
Paige Prostate Detect was the first FDA-authorized AI in pathology, flagging suspicious foci on prostate core biopsies. It received class II designation through the agency’s de novo classification pathway in 2021. In April 2025, Paige PanCancer Detect received breakthrough device designation for detecting suspicious foci across multiple tissues and organs — the first such designation for a multitissue AI assist. Tempus acquired Paige in August 2025, merging approximately 7 million slides with multiomics data to accelerate the path from discovery to regulated tools.
Ibex Prostate Detect, an AI-powered cancer diagnostics from Galen Second Read, received FDA 510(k) clearance in February 2025. This tool flags cases initially signed out benign for re-review, producing case-level alerts and heatmaps of likely cancer regions — a pragmatic backstop in high-volume services.
These are assistive tools, not autonomous diagnosis. For oncologists, their value lies in reducing false negatives, standardizing triage, and supporting workforce-constrained pathology services.
Virtual Staining: Histology to Sequencing
Virtual staining represents a broader shift in oncology AI, from tools that primarily detect cancer on slides to systems that actively improve downstream clinical workflows, including molecular testing, tissue utilization, and treatment planning.
ClearStain (Pictor Labs; research use in the US) generates a hematoxylin and eosin (H&E)-equivalent digital image from an unstained section destined for molecular testing. Pathologists can annotate tumor on the same section that undergoes DNA/RNA extraction, what you see is what you sequence. Proscia is integrating Pictor’s virtual stains into its Concentriq platform, signaling ecosystem uptake.
The implications are significant. This approach preserves tissue in small biopsies, improves tumor purity selection, reduces failure of next-generation sequencing (NGS) due to tissue depletion, and shortens turnaround time. For tissue-limited lung or prostate rebiopsies, this is immediately relevant. The conceptual shift: The diagnostic slide becomes an active guide for downstream molecular workflows.
Multimodal Models: What Comes Next
Companies such as Noetik are training multimodal transformer models (OCTO/OCTO-VirtualCell) on paired H&E slides, spatial transcriptomics, spatial proteomics, and sequencing data from thousands of tumors (nearly 40 million cells). These systems simulate spatial single-cell gene expression in context.
They are not FDA diagnostics yet, but this is the pipeline for future computational biomarkers. Partnerships, such as those with Agenus, aim to derive predictive biomarkers for immunotherapy directly from morphology plus learned biology. The trajectory is clear: Detection, prognosis, treatment prediction, and pathway inference are all derived from the diagnostic slide.
The Moravec Paradox in Pathology
The Moravec paradox states that what is easy for humans is often hard for AI, and what is hard for humans may be easy for AI. In digital pathology, that dynamic is visible:
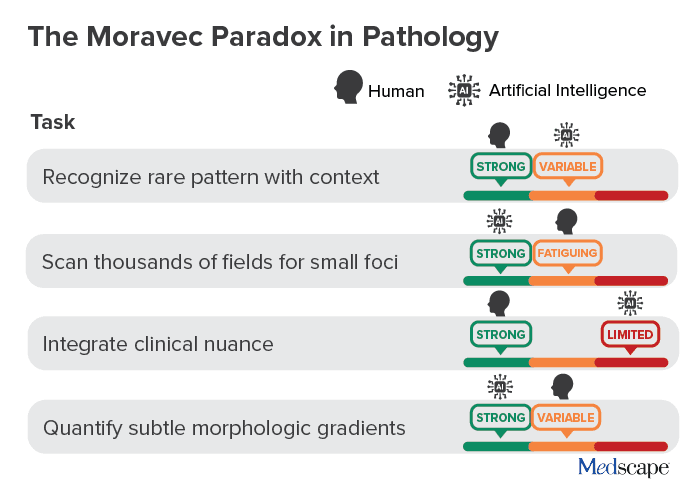
AI excels at large-scale scanning and quantification. Humans excel at contextual reasoning and clinical integration. The future is not replacement but division of labor.
What Oncologists Should Do Now
Confirm whole-slide imaging readiness. Ask your pathology group whether primary diagnosis has been validated on the current whole-slide imaging platform according to College of American Pathologists guidance, including appropriate case mix and concordance. As a practical benchmark, many programs cite at least 60 cases and more than 95% concordance.
Also ask how performance is monitored over time, particularly when scanners, stains, or viewing software change.
Then ask the key operational question: Can your current viewer support third-party AI modules? This determines whether you can adopt one tool now and scale later without rebuilding infrastructure.
Clarify remote review policy. Ensure that your pathology and compliance teams have a clearly defined operational policy for remote digital review. Under current CLIA guidance, remote review of digital slides is permitted under the primary CLIA certificate when required conditions are met. Remote cytology digital review requires separate certification after March 23, 2026, and physical glass slides cannot be reviewed remotely under the primary certificate.
For oncology teams, this matters because digital workflows can expand access to subspecialty expertise across sites, but only if implemented within a compliant framework.
Build a prostate decision pathway. If you treat localized prostate cancer, determine whether ArteraAI ordering is enabled and incorporate it into multidisciplinary discussions when intensification decisions are on the table. Set expectations that this is an AI-enabled biomarker augmenting clinico-pathologic risk and genomics — not replacing them. Note Medicare payment status in your pathway to reduce billing friction.
Pilot virtual staining where tissue is scarce. For NGS-bound specimens, ClearStain can preserve tissue and reduce sequencing failures. Align on indications, such as scant core biopsies, along with validation steps and appropriate reporting language. In the United States, these tools remain research use only unless locally validated.
What to Watch Next
Expect a trickle-down effect from foundational-model class encoders, such as PLUTO-4, which will boost accuracy and generalization across multiple commercial tools — backbone upgrades you may not see named in reports. Pan-organ detection is also on the near-term horizon. FDA breakthrough designation for Paige PanCancer Detect suggests a “universal triage” across organ systems. Additionally, health-system impact data will soon become widely available through the National Health Service Vanguard Path program, which is set to provide prospective evidence for AI-guided treatment selection in routine practice.
Bottom Line
Digital pathology is no longer about viewing slides on a screen. It is about extracting additional biologic signal from tissue already collected, without another biopsy.
When evaluating any AI tool, consider these three questions:
- What is the regulatory status: de novo, 510(k), or research use?
- Is it validated on our scanners and viewers?
- Does it change a treatment decision I make every week?
If the answer to the third question is yes, it belongs in the multidisciplinary conversation. The microscope isn’t disappearing. It’s becoming computational.
The next chapter will not be about whether AI can detect cancer; that question is largely settled. It will be about whether image-derived biomarkers can reliably guide systemic therapy, immunotherapy selection, and resistance monitoring across tumor types. As foundation models mature and more slide-based predictors enter regulated use, the diagnostic biopsy will increasingly function as a multiomic sensor with morphology, molecular inference, and treatment guidance layered into a single workflow. Our job as oncologists is not to chase every tool, but to integrate the ones that meaningfully improve decisions for the patient in front of us.
Thoughts? Drop me a line at Arturo.AI.MedTech@gmail.com or DM on X (@DrArturoAI. Let’s keep the conversation — and the foundational models — going forward.
Arturo Loaiza-Bonilla, MD, MSEd, is the co-founder and chief medical AI officer at Massive Bio, a company connecting patients to clinical trials using artificial intelligence. His research and professional interests focus on precision medicine, clinical trial design, digital health, entrepreneurship, and patient advocacy. Dr Loaiza-Bonilla serves as systemwide chief of hematology and oncology at St. Luke’s University Health Network, where he maintains a connection to patient care by attending to patients 2 days a week.
No comments:
Post a Comment
Note: Only a member of this blog may post a comment.